Page 29 - AN-2-2
P. 29
Advanced Neurology Seizures and CKD
In CKD, there can be disruptions to extracellular ion several proposed mechanisms. The direct and indirect toxic
homeostasis and altered protein-energy metabolism, which effects of uremia have been described in the previous studies.
can potentially lead to neuronal membrane instability Urea is a source of cyanate, carbamylated compounds, and
(Figure 1) [20,21] . There are several proposed etiologies that ammonia accumulation . Accumulation of guanidine,
[26]
could explain CKD-associated provoked seizures. These guanidinosuccinic acid and methyl-guanidine, and
include uremic encephalopathy; AKI-on-CKD seizures; other compounds have also been identified as potential
seizures caused by electrolyte and blood sugar disturbances; neurotoxins in uremic encephalopathy; the increased efflux
seizures in the context of uncontrolled hypertension, of these compounds have a stimulatory effect on N-methyl-
including posterior reversible encephalopathy syndrome D-aspartate (NMDA), glutamate receptors (GluR), and an
(PRES); erythropoietin and medication-induced seizures inhibitory effect on GABA receptors [27,28] . These neurotoxins
in the dialysis and post-transplantation population; and contribute to the imbalance of excitatory and inhibitory
cerebrovascular event-related seizures. In this section, we neurotransmitters, as well as deregulation of transporters in
will discuss the different CKD scenarios which can lead to the blood-brain barrier involved in neuronal degeneration
potential instability of the neuronal membrane resulting in and vascular inflammatory pathways [16,27] .
seizure activity.
2.2. AKI-on-CKD seizures
2.1. Uremic encephalopathy AKI is commonly observed in critically unwell patients with
Uremic encephalopathy is an acute syndrome which is remote organ dysfunction. The CNS is vulnerable following
observed in uremia . The blood-brain barrier is a unique acute kidney dysfunction, particularly in patients with pre-
[22]
structure that maintains cerebral homeostasis by acting as existing CKD [29,30] . In AKI-on-CKD, tubular epithelial cells
a shield against circulating toxins and pro-inflammatory undergoing apoptosis trigger a profound inflammatory
molecules . This homeostatic function is disrupted when the reaction, disrupting the osmolality, acid-base, and water-
[23]
central nervous system (CNS) is subject to constant systemic electrolyte balances . There is an increased accumulation
[31]
inflammatory assault [23-25] . The exact pathophysiological of urea, creatinine, guanidine, homocysteine, dimethyl
basis of uremic encephalopathy is uncertain, but there are arginine and reactive oxygen species, all of which are
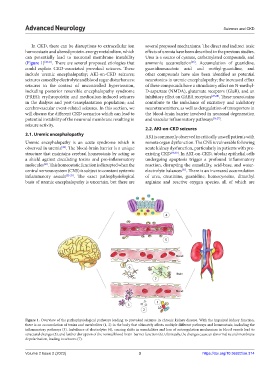
Figure 1. Overview of the pathophysiological pathways leading to provoked seizures in chronic kidney disease. With the impaired kidney function,
there is an accumulation of toxins and metabolites (1, 2) in the body that ultimately affects multiple different pathways and homeostasis, including the
inflammatory pathways (3). Imbalance of electrolytes (4), causing shifts in osmolalities and loss of autoregulation mechanism in blood vessels lead to
structural changes (5), and further disruption of the normal blood-brain-barrier function (6). Ultimately, the changes cause an abnormal neural membrane
depolarization, leading to seizures (7).
https://doi.org/10.36922/an.314
Volume 2 Issue 2 (2023) olume 2 Issue 2 (2023) 3 3 https://doi.org/10.36922/an.314
V