Page 109 - JCBP-3-3
P. 109
Journal of Clinical and
Basic Psychosomatics Comorbid MDD and FMS
symptoms. Laboratory tests and imaging studies, including The final diagnosis was recurrent severe MDD and
electrocardiogram, spot electroencephalogram, cardiac and FMS. Thus, she was administered pregabalin (75 mg twice
abdominal ultrasound, and chest computed tomography, daily), venlafaxine extended-release (225 mg once daily),
10
revealed no significant abnormalities. Brain magnetic and lorazepam (0.5 mg at bedtime). By day 12, the patient
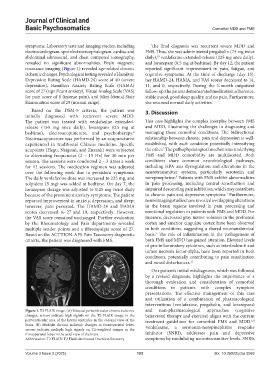
resonance imaging (Figure 1) revealed age-related chronic reported significant improvement in pain, fatigue, and
ischemic changes. Psychological testing revealed a Hamilton cognitive symptoms. At the time of discharge (day 15),
Depression Rating Scale (HAMD-24) score of 40 (severe her HAMD-24, HAMA, and VAS scores decreased to 14,
depression), Hamilton Anxiety Rating Scale (HAMA) 11, and 0, respectively. During the 3-month outpatient
score of 27 (significant anxiety), Visual Analog Scale (VAS) follow-up, the patient demonstrated medication adherence,
for pain score of 8 (intense pain), and Mini-Mental State stable mood, good sleep quality, and no pain. Furthermore,
Examination score of 29 (normal range). she resumed normal daily activities.
Based on the DSM-5 criteria, the patient was 3. Discussion
initially diagnosed with recurrent severe MDD.
The patient was treated with venlafaxine extended- This case highlights the complex interplay between FMS
release (150 mg once daily), lorazepam (0.5 mg at and MDD, illustrating the challenges in diagnosing and
bedtime), electroacupuncture, and psychotherapy. managing these comorbid conditions. The bidirectional
9
Electroacupuncture was performed by an acupuncturist relationship between chronic pain and depression is well-
experienced in traditional Chinese medicine. Specific established, with each condition potentially intensifying
3
acupoints (Hegu, Neiguan, and Zusanli) were subjected the other. The pathophysiological mechanisms underlying
to alternating frequencies (2 – 15 Hz) for 30 min per FMS and MDD comorbidity are multifaceted. Both
session. The sessions were conducted 2 – 3 times a week conditions share common neurobiological pathways,
for 12 sessions. The medication regimen was adjusted including HPA axis dysregulation and alterations in the
over the following week due to persistent symptoms. neurotransmitter systems, particularly serotonin and
The daily venlafaxine dose was increased to 225 mg, and norepinephrine. Patients with FMS exhibit abnormalities
5
zolpidem (5 mg) was added at bedtime. On day 7, the in pain processing, including central sensitization and
lorazepam dosage was adjusted to 0.25 mg twice daily impaired descending pain inhibition, which may contribute
because of the prominent anxiety symptoms. The patient to chronic pain and depressive symptoms. Furthermore,
1
reported improvement in anxiety, depression, and sleep; neuroimaging studies have revealed overlapping alterations
however, pain persisted. The HAMD-24 and HAMA in the brain regions involved in pain processing and
scores decreased to 27 and 10, respectively. However, emotional regulation in patients with FMS and MDD. For
the VAS score remained unchanged. Further evaluation instance, decreased gray matter volumes in the prefrontal
by the Rheumatology and Pain departments revealed cortex and anterior cingulate cortex have been observed
multiple tender points and a fibromyalgia score of 27. in both conditions, suggesting a shared neuroanatomical
3
Based on the ACTTION-APS Pain Taxonomy diagnostic basis. The role of inflammation in the pathogenesis of
criteria, the patient was diagnosed with FMS. both FMS and MDD has gained attention. Elevated levels
of pro-inflammatory cytokines, such as interleukin-6 and
tumor necrosis factor-alpha, have been reported in both
conditions, potentially contributing to pain sensitization
and mood disturbances. 11
Our patient’s initial misdiagnosis, which was followed
by a revised diagnosis, highlights the importance of a
thorough evaluation and consideration of comorbid
conditions in patients with complex symptom
presentations. The effective management of this case
and utilization of a combination of pharmacological
interventions (venlafaxine, pregabalin, and lorazepam)
Figure 1. T2 FLAIR image. (A) Bilateral periventricular chronic ischemic and non-pharmacological approaches (cognitive
changes, arrows indicate high signals on the T2-FLAIR image in the behavioral therapy and exercise) aligns with the current
periventricular area of the lateral ventricles in the coronal view of the treatment guidelines for comorbid FMS and MDD.
6,8
brain. (B) Multiple chronic ischemic changes in frontoparietal lobes, Venlafaxine, a serotonin-norepinephrine reuptake
arrows indicate multiple high signals on T2-weighted images in the
frontoparietal lobes in the axial view of the brain. inhibitor (SNRI), addresses pain and depressive
Abbreviation: T2 FLAIR: T2 Fluid-Attenuated Inversion Recovery symptoms by modulating neurotransmitter levels. SNRIs
Volume 3 Issue 3 (2025) 103 doi: 10.36922/jcbp.5069