Page 108 - AN-4-4
P. 108
Advanced Neurology Incidental PFBC: Case report
of individuals with autosomal dominant PFBC remain
asymptomatic throughout their lifetime. 2
To date, mutations in six genes have been found to
be associated with PFBC; four are autosomal dominant
(solute carrier family 20 member 2, xenotropic and
polytropic retrovirus receptor 1, platelet-derived growth
factor beta [PDGFB], and platelet-derived growth factor
receptor beta [PDGFRB]) and two are autosomal recessive
(myogenesis regulating glycosidase and junctional-
adhesion molecule-2). 2
Herein, we report an asymptomatic case of PFBC
discovered incidentally on a computed tomography (CT)
scan of the brain in a middle-aged male being evaluated
for sinus complaints. Comprehensive family history and
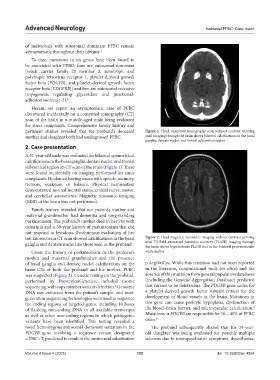
pertinent studies revealed that the proband’s deceased Figure 1. Head computed tomography scan without contrast utilizing
mother and daughter both had undiagnosed PFBC. axial imaging through the brain shows bilateral calcifications in the basal
ganglia, dentate nuclei, and frontal subcortical region
2. Case presentation
A 51-year-old male was evaluated for bilateral symmetrical
calcifications in the basal ganglia, dentate nuclei, and frontal
subcortical region on CT scan of the brain (Figure 1). These
were found incidentally on imaging performed for sinus
complaints. He denied having issues with speech, memory,
tremors, weakness, or balance. Physical examination
demonstrated normal mental status, cranial nerve, motor,
and cerebellar assessments. Magnetic resonance imaging
(MRI) of the brain was not performed.
Family history revealed that our patient’s mother and
maternal grandmother had dementia and long-standing
parkinsonism. The proband’s mother died in her 70s with
dementia and a 30-year history of parkinsonism that did
not respond to levodopa. Posthumous evaluation of her
last known brain CT scan showed calcifications in the basal Figure 2. Head magnetic resonance imaging without contrast utilizing
ganglia and dentate nuclei like those seen in the proband. axial T2 fluid-attenuated inversion recovery (FLAIR) imaging through
the brain shows hyperintense FLAIR foci in the bilateral periventricular
Given the history of parkinsonism in the proband’s white matter
mother and maternal grandmother and the presence
of basal ganglia and dentate nuclei calcifications on the p.Arg100Cys. While this mutation had not been reported
brain CTs of both the proband and his mother, PFBC in the literature, computational tools (in silico) and the
was suspected (Figure 2). Genetic testing on the proband, absence of this mutation from general population databases
performed by PreventionGenetics, included exome (including the Genome Aggregation Database) predicted
sequencing with copy number variant detection. Genomic this variant to be deleterious. The PDGFB gene codes for
4
DNA was extracted from the patient’s sample, and next- a platelet-derived growth factor subunit critical for the
generation sequencing technologies were used to sequence development of blood vessels in the brain. Mutations in
the coding regions of targeted genes, including 10 bases this gene can cause pericyte hypoplasia, dysfunction of
5
of flanking non-coding DNA in all available transcripts the blood–brain barrier, and microvascular calcification.
as well as other non-coding regions in which pathogenic Mutations in PDGFB are responsible for 10 – 40% of PFBC
6
variants have been identified. This testing revealed a cases.
novel heterozygous autosomal dominant mutation in the The proband subsequently shared that his 24-year-
PDGFB gene involving a sequence variant designated old daughter was being evaluated for possible multiple
c.298C>T, predicted to result in the amino acid substitution sclerosis due to neuropsychiatric symptoms, dysesthesias,
Volume 4 Issue 4 (2025) 102 doi: 10.36922/an.4854