Page 115 - AN-4-4
P. 115
Advanced Neurology CAA-related inflammatory case
A B the risks of bleeding, infection, and aggravation of the
condition caused by invasive examinations, as well as the
poor acceptance of patients, the current utilization rate of
brain biopsy is only 0.5 – 1%. In terms of genes, being a
6
homozygous ε4/ε4 in the ApoE-ε4 allele is considered the
only definite risk factor for CAA-ri. The carriage rate of ε4/
7
ε4 in CAA-ri patients is 76.9%, whereas only 5.1% of non-
ICAA patients carry the homozygous gene. Nevertheless,
8
this patient did not undergo complete genetic testing. In
addition, high-titer anti-Aβ autoantibodies can be detected
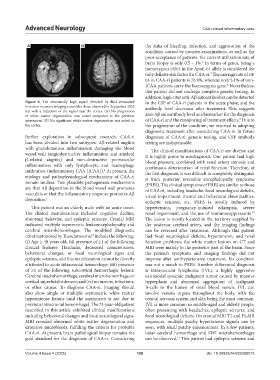
Figure 6. The abnormally high signal detected by fluid-attenuated in the CSF of CAA-ri patients in the acute phase, and the
inversion recovery imaging resembles those observed in September 2023 antibody level decreases after treatment. This suggests
but with a reduction of the signal near the cortex. (A) No progression
of white matter degeneration was noted compared to the previous anti-Aβ autoantibody level as a biomarker for the diagnosis
9
assessment. (B) No significant white matter degeneration was noted in of CAA-ri and the monitoring of treatment effects. Due to
the cortex. the progression of the condition, we resorted to adopting
diagnostic treatment after considering CAA-ri. In future
further exploration in subsequent research, CAA-ri diagnoses of CAA-ri, genetic testing, and CSF antibody
has been divided into two subtypes: Aβ-related angiitis testing are indispensable.
with granulomatous inflammation damaging the blood The clinical manifestations of CAA-ri are diverse and
vessel wall (angiodestructive inflammation and amyloid it is highly prone to misdiagnosis. Our patient had high
β-related angiitis) and non-destructive perivascular blood pressure, combined with renal artery stenosis and
inflammation with only lymphocyte and macrophage continuous deterioration of renal function. Therefore, at
infiltration (inflammatory CAA [ICAA]). At present, the the first diagnosis, it was difficult to completely distinguish
4
etiology and pathophysiological mechanisms of CAA-ri it from posterior reversible encephalopathy syndrome
remain unclear. Two plausible pathogenesis mechanisms (PRES). The clinical symptoms of PRES are similar to those
are that Aβ deposition in the blood vessel wall promotes of CAA-ri, including headache, focal neurological deficits,
vasculitis or that the inflammatory response promotes Aβ visual impairment, mental and behavioral abnormalities,
deposition. 1
epileptic seizures, etc. PRES is usually induced by
This patient was an elderly male with an acute onset. hypertension, pregnancy-induced eclampsia, severe
The clinical manifestations included cognitive decline, renal impairment, and the use of immunosuppressants.
10
abnormal behavior, and epileptic seizures. Cranial MRI The lesion is mostly located in the territory supplied by
indicated multiple asymmetric leukoencephalopathy and the posterior cerebral artery, and the imaging findings
cerebral microhemorrhages. The modified diagnostic can be reversed after treatment. Although this patient
criteria proposed by Theodorou et al. include the following: had focal neurological deficits, hypertension, and renal
5
(i) Age ≥ 40 years old; (ii) presence of ≥1 of the following function problems, the white matter lesions on CT and
clinical features: Headache, decreased consciousness, MRI were mainly in the posterior part of the brain. Since
behavioral changes, or focal neurological signs and the patient’s symptoms and imaging findings did not
epileptic seizures, and this manifestation cannot be directly improve after antihypertensive treatment, his condition
attributed to acute intracranial hemorrhage; (iii) presence was not a match to PRES. Another differential diagnosis
of ≥1 of the following subcortical hemorrhagic lesions: is intravascular lymphoma (IVL), a highly aggressive
Cerebral macrohemorrhage, cerebral microhemorrhage, or extranodal systemic malignant tumor caused by massive
cortical superficial siderosis; and (iv) no tumors, infections, hyperplasia and abnormal aggregation of malignant
or other causes. To diagnose CAA-ri, imaging should B-cells in the lumen of small blood vessels. IVL can
also show single or multiple asymmetric white matter involve various organs throughout the body, with the
hyperintense lesions (and the asymmetry is not due to central nervous system and skin being the most common.
previous intracranial hemorrhage). The 73-year-old patient IVL is more common in middle-aged and elderly people,
described in this article exhibited clinical manifestations often presenting with headaches, epileptic seizures, and
including behavioral changes and focal neurological signs. focal neurological deficits. On cranial MRI T2 and FLAIR
MRI revealed abnormal white matter degeneration and sequences, multiple patchy hyperintense signals can be
extensive microbleeds, fulfilling the criteria for probable seen, with small patchy enhancement. In a few patients,
CAA-ri. At present, brain pathological biopsy remains the lobar cerebral hemorrhage and SWI microhemorrhages
gold standard for the diagnosis of CAA-ri. Considering can be observed. This patient had epileptic seizures and
11
Volume 4 Issue 4 (2025) 109 doi: 10.36922/AN025080015