Page 45 - AN-4-4
P. 45
Advanced Neurology Lipid metabolism and Parkinson’s disease
PE/PS transport, impairing CL synthesis, and triggering promotes α-Syn aggregation; 96,97 and GlcCer accumulation
mitochondrial apoptosis. GBA1 mutations (a major triggers a “lysosomal damage–α-Syn aggregation–
29
PD risk factor) reduce GCase activity, causing GlcCer neuroinflammation” cycle. Furthermore, SCFAs link gut
107
accumulation in lysosomes. GlcCer inhibits the V-ATPase dysbiosis to neuroinflammation through TLR4/NF-κB
proton pump, raising lysosomal pH, inactivating LAL, and activation, while LPC imbalance exacerbates inflammation
impairing lipophagy while disrupting α-Syn’s tetramer– and α-Syn pathology through GPR35–ERK signaling. In
131
26
monomer balance to promote aggregation. In addition, addition, as discussed in Section 3.3.2, gangliosides GM1 and
LPC activates the GPR35–ERK pathway, disrupting Golgi GM3 play pivotal roles in PD pathogenesis, with GM1 acting
structure and blocking GCase transport to lysosomes, as a neuroprotective agent and GM3 promoting pathological
exacerbating GlcCer accumulation. 131 progression. 116-118 Disruptions in their expression levels or
Lipid metabolism dysregulation involves multiple metabolic equilibrium directly contribute to dopaminergic
pathogenic pathways: FA imbalance (e.g., PUFA reduction and neuron loss and abnormal α-Syn aggregation, thereby
SCFA-mediated inflammation), phospholipid remodeling accelerating disease progression.
(e.g., CL/PC/PE dysfunction), sphingolipid disruption (e.g., Despite these insights, key questions remain: the
Cer/GlcCer accumulation), and cholesterol metabolite- causal timeline of lipid abnormalities (whether initiator
driven α-Syn aggregation. 59,144-146 These abnormalities form a or consequence of PD) requires longitudinal validation,
complex network: reduced PUFAs impair membrane fluidity and cell-type specificity in lipid metabolism (e.g., neuronal
and neuroprotection; CL deficiency disrupts mitochondrial vs. glial differences) needs elucidation through single-
respiration and α-Syn stability; 88,145 PC/PE imbalance cell approaches. Translational challenges, such as BBB
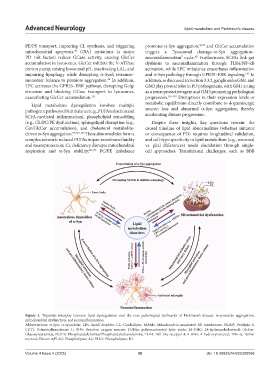
Figure 1. Tripartite interplay between lipid dysregulation and the core pathological hallmarks of Parkinson’s disease: α-synuclein aggregation,
mitochondrial dysfunction, and neuroinflammation
Abbreviations: α-Syn: α-synuclein; LDs: Lipid droplets; CL: Cardiolipin; MAMs: Mitochondria-associated ER membranes; PLIN5: Perilipin-5;
CPT1: Palmitoyltransferase 1; ROS: Reactive oxygen species; PUFAs: polyunsaturated fatty acids; 24-OHC: 24-hydroxycholesterol; GlcCer:
Glucosylceramide; PC/PE: Phosphatidylcholine/Phosphatidylethanolamine; TLR4: Toll-like receptor 4; 4-HNE: 4-hydroxynonenal; TNF-α: Tumor
necrosis FActor-α;PLA2: Phospholipase A2; PLD1: Phospholipase D1.
Volume 4 Issue 4 (2025) 39 doi: 10.36922/AN025320086