Page 11 - GHES-3-3
P. 11
Global Health Economics and
Sustainability
Sustainable therapeutic Artemisia
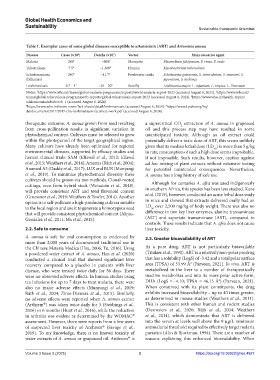
Table 1. Exemplar cases of some global diseases susceptible to artemisinin (ART) and Artemisia annua
Disease Cases (×10 ) Deaths (×10 ) Vector Main causative agent
6
3
Malaria 249 1 ~600 1 Mosquito Plasmodium falciparum, P. vivax, P. ovale
Tuberculosis 7.5 2 ~1,300 2 Human Mycobacterium tuberculosis
Schistosomiasis 240 3 ~11.7 3 Freshwater snails Schistosoma guineensis, S. intercalatum, S. mansoni, S.
(bilharzia) japonicum, S. mekongi
Leishmaniasis 0.7 – 1 4 ~20 – 30 5 Sandfly Leishmania major, L. infantum, L. tropica, L. Donovani
Notes: https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2023 (accessed August 6, 2024). https://www.who.int/
1
2
3
teams/global-tuberculosis-programme/tb-reports/global-tuberculosis-report-2023 (accessed August 6, 2024). https://www.who.int/health-topics/
schistosomiasis#tab=tab_1 (accessed August 6, 2024)
5
4 https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed August 6, 2024). https://www3.paho.org/hq/
dmdocuments/2017/2017-cha-leishmaniasis-factsheet-work.pdf (accessed August 6, 2024).
therapeutic outcome. A. annua grown from seed resulting a supercritical CO extraction of A. annua in grapeseed
2
from cross-pollination results in significant variation in oil and this process step may have resulted in some
phytochemical content. Cultivars must be selected to grow unanticipated toxicity. Although an oil extract could
within the photoperiod of the target geographical region. potentially deliver a toxic dose of ART, this seems unlikely
Many cultivars have already been optimized for regional given that its median lethal dose (LD ) is more than 5 g/kg
50
environmental climates, supported by efficacy studies and in rats; consumption of such a high dose seems improbable,
human clinical trials: SAM (Elfawal et al., 2012; Elfawal if not impossible. Such results, however, caution against
et al., 2015; Weathers et al., 2014), Artemis (Räth et al., 2004); ad hoc mixing of plant extracts without extensive testing
Anamed A3 (Daddy et al., 2017), LUX and BUR (Munyangi for potential unintended consequences. Nevertheless,
et al., 2019). To minimize phytochemical diversity these A. annua has a long history of safe use.
cultivars should be grown via two methods. Clonal-rooted
cuttings, even from hybrid stock (Wetzstein et al., 2018), Although for centuries A. afra was used indigenously
will provide consistent ART and total flavonoid content in southern Africa, this species has been less studied. Kane
(Gruessner et al., 2019; Weathers & Towler, 2014). Another et al. (2019), however, conducted an acute lethal dose study
option is to self-pollinate a high-producing cultivar suitable in mice and showed that extracts delivered orally had an
to the local region and use it to generate a homozygous seed LD over 2,500 mg/kg of body weight. There was also no
50
that will provide consistent phytochemical content (Alejos- difference in two key liver enzymes, alanine transaminase
Gonzalez et al., 2011; Ma et al., 2015). (ALT) and aspartate transaminase (AST), compared to
controls. These results indicate that A. afra does not cause
2.2. Safe to consume liver toxicity.
A. annua is safe for oral consumption as evidenced by 2.3. Greater bioavailability of ART
more than 2,000 years of documented traditional use in
the Chinese Materia Medica (Hsu, 2006; Tu, 2016). Using As a pure drug, ART is not particularly bioavailable
a powdered water extract of A. annua, Han et al. (2020) (Titulaer et al., 1990). ART is a relatively non-polar prodrug
conducted a clinical trial that showed significant liver that has a solubility (LogS) of -3.42 and a total polar surface
2
recovery compared to a placebo in patients with liver area (TPSA) of 53.99 Å (Parveen, 2021). In vivo, ART is
disease, who were treated twice daily for 56 days. There metabolized in the liver to a number of therapeutically
were no observed adverse effects. In human studies using inactive metabolites and into its more polar active form,
2
tea infusions for up to 7 days to treat malaria, there were DHA (LogS = -4.10; TPSA = 46.15 Å ) (Parveen, 2021).
also no major adverse effects (Munyangi et al., 2019; When consumed with its plant constituents, the drug
Räth et al., 2004; Zime-Diawara et al., 2015). Similarly, exhibits increased bioavailability – up to 40 times greater,
no adverse effects were reported when A. annua extract as determined in mouse studies (Weathers et al., 2011).
(Arthrem ) was taken twice daily for 3 (Stebbings et al., This is consistent with other human and rodent studies
2016) or 6 months (Hunt et al., 2016), while the reduction (Desrosiers et al., 2020; Räth et al., 2004; Weathers
in arthritis was evident as determined by the WOMAC et al., 2014), which demonstrate that ART is delivered
assessment. However, there were reports from a few users into the serum at levels well above the 9 µg/L minimum
of suspected liver toxicity of Anthrem (Savage et al., antimalarial threshold required to effectively target malaria
2019). To my knowledge, there is no known toxicity of parasites (Alin & Bjorkman, 1994). There are a number of
water extracts of A. annua or grapeseed oil. Anthrem is reasons explaining this enhanced bioavailability. When
Volume 3 Issue 3 (2025) 3 https://doi.org/10.36922/ghes.4927