Page 33 - GHES-3-3
P. 33
Global Health Economics and
Sustainability
Semaglutide for treating T2D and obesity
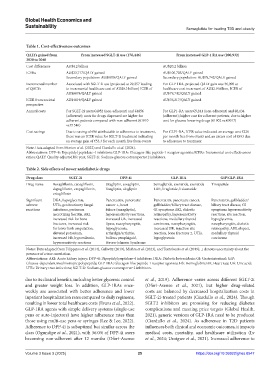
Table 1. Cost‑effectiveness outcomes
QALYs gained from From increased SGLT‑2i use (176,446) From increased GLP‑1 RA use (200,932)
2020 to 2040
Cost differences AU$4.2 billion AU$20.2 billion
ICERs AU$23,717/QALY gained AU$100,705/QALY gained
Secondary population: AU$8878/QALY gained Secondary population: AU$79,742/QALY gained
Incremental number Associated with SGLT-2i use (projected at 29,357 leading For GLP-1RA, projected QALY gain was 36,090 at
of QALYs to incremental healthcare cost of AU$0.3 billion) ICER of healthcare cost increment of AU$2.9 billion, ICER of
AU$8878/QALY gained AU$79,742/QALY gained
ICER from societal AU$4819/QALY gained AU$76,217/QALY gained
perspective
Annual costs For SGLT-2i users:€4952 (non-adherent) and €4856 For GLP1-RA users:€5,241 (non-adherent) and €6,134
(adherent); costs for drugs dispensed are higher for (adherent); higher cost for adherent patients, due to higher
adherent patients compared with non-adherent (€1910 cost for glucose-lowering drugs (€1902 vs.€3017)
vs.€1546)
Cost savings Due to saving of €96 attributable to adherence to treatment, For GLP1-RA, ICER value indicated an average cost €228
there was an ICER value for SGLT-2i treatment indicating per month free from events and an excess cost of €893 due
an average gain of €53.3 for each month free from events to adherence to treatment
Note: Data adapted from Morton et al. (2022) and Ciardullo et al. (2024).
Abbreviations: DPP-4i: Dipeptidyl peptidase-4 inhibitors; GLP-1RA: Glucagon-like peptide-1 receptor agonists; ICERs: Incremental cost-effectiveness
ratios; QALY: Quality-adjusted life year; SGLT-2i: Sodium-glucose cotransporter 2 inhibitors.
Table 2. Side effects of newer antidiabetic drugs
Drug class SGLT‑2i DPP‑4i GLP‑1RA GIP/GLP‑1RA
Drug name Bexagliflozin, canagliflozin, Sitagliptin, saxagliptin, Semaglutide, exenatide, exenatide Tirzepatide
dapagliflozin, empagliflozin, linagliptin, alogliptin LAR, liraglutide, lixisenatide
ertugliflozin
Significant DKA, hypoglycemia, Pancreatitis, pancreatic Pancreatitis, pancreatic cancer, Pancreatitis, gallbladder/
adverse UTIs, genitourinary fungal cancer ±, heart gallbladder/biliary tract disease, biliary tract disease, GI
reactions infections, perineum failure (saxagliptin), GI symptoms AKI, diabetic symptoms hypersensitivity
necrotizing fasciitis, AKI, hypersensitivity reactions, retinopathy, hypersensitivity reactions, site reaction,
increased risk for bone increased UA, increased reactions, medullary thyroid hypoglycemia,
fractures, increased risk lipase, nasopharyngitis, carcinoma, nasopharyngitis, nasopharyngitis, diabetic
for lower limb amputation, hypoglycemia, increased HR, injection site retinopathy, AKI, alopeci,
elevated potassium, arthralgia/arthritis, reaction, bone fractures ±, UTIs, medullary thyroid
increased Hb, hypovolemia, bullous pemphigoid, hypoglycemia carcinoma
hypersensitivity reactions Steven-Johnson Syndrome
Notes: Data adapted from Filippatos et al. (2014), Gallwitz (2019), Mishra et al. (2023), and Tentolouris et al. (2019); ± denotes uncertainty about the
presence of a true correlation.
Abbreviations: AKI: Acute kidney injury; DPP-4i: Dipeptidyl peptidase-4 inhibitors; DKA: Diabetic ketoacidosis; GI: Gastrointestinal; GIP:
Glucose-dependent insulinotropic polypeptide; GLP-1RA: Glucagon-like peptide-1 receptor agonists; Hb: Hemoglobin; HR: Heart rate; UA: Uric acid;
UTIs: Urinary tract infections; SGLT-2i: Sodium-glucose cotransporter 2 inhibitors.
due to its clinical benefits, including better glycemic control et al., 2019). Adherence varies across different SGLT-2i
and greater weight loss. In addition, GLP-1RAs once- (Ofori‐Asenso et al., 2021), but higher drug-related
weekly are associated with better adherence and lower costs are balanced by decreased hospitalization costs in
inpatient hospitalization rates compared to daily regimens, SGLT-2i-treated patients (Ciardullo et al., 2024). Though
resulting in lower total healthcare costs (Evans et al., 2022). SGLT2 inhibitors are promising for reducing diabetes
GLP-1RA agents with simple delivery systems (single-use complications and meeting price targets (Global Health,
pens or auto-injectors) have higher adherence rates than 2021), generic versions of GLP-1RA need to be produced
those using multi-use pens or syringes (Lee & Lee, 2022). (Ciardullo et al., 2024). As adherence in T2D patients
Adherence to DPP-4i is suboptimal but similar across the influences both clinical and economic outcomes, it impacts
class (Ogundipe et al., 2021), with 36.0% of DPP-4i users medical costs, mortality, and healthcare utilization (Lv
becoming non-adherent after 12 months (Ofori-Asenso et al., 2024; Uzoigwe et al., 2021). Increased adherence to
Volume 3 Issue 3 (2025) 25 https://doi.org/10.36922/ghes.8547