Page 126 - IMO-1-1
P. 126
Innovative Medicines & Omics Promising molecule against SARS-CoV-2 nucleocapsid
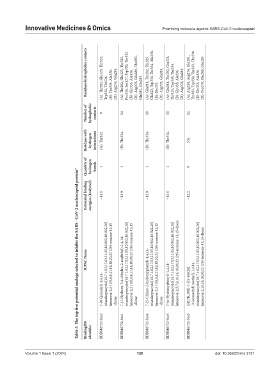
Residues‑hydrophobic contacts (A): Thr282, Glu323, Thr325, Trp330, Thr334. (B): Thr334, Ala336. (D): Arg276, Gln283. (A): Thr282, Glu323, Thr325, Pro326, Ser327, Trp330, Thr332. (B): Gly335, Ala336. (D): Arg276, Glu280, Thr282, Gln281, Gln283. (A): Gln281, Thr282, Thr325, Glu323, Trp330, Thr334, Ala336. (B): Gly335. (D): Arg276, Gln283. (A): Gln281, Thr282, Glu323, Thr325, Trp330, Thr334. (B): Gly335, Ala336. (D)
Number of hydrophobic contacts 9 14 10 10 12
Residues with hydrogen interactions (A): Thr332. (B): Thr334. (B): Thr334. (B): Thr334. NA
Quantity of hydrogen bonds 1 1 1 1 0
Table 3. The top five potential analogs selected to inhibit the SARS ‑ CoV‑2 nucleocapsid protein*
Estimated binding energies (kcal/mol) −14.1 −13.9 −12.9 −12.6 −12.2
IUPAC Name triazaheptacyclo[16.7.1.02,17.03,11.05,10.012,16.022,26] hexacosa-2,5 (10),6,8,11,16,18,20,22 (26)-nonane-13,15 7-(1-Hydroxy-3,4-dihydro-2-naphthyl)-1,4,14- triazaheptacyclo[16.7.1.02,17.03,11.05,10.012,16.022,26] hexacosa-2,5 (10),6,8,11,16,18,20,22 (26)-nonane-13,15 triazaheptacyclo[16.7.1.02,17.03,11.05,10.012,16.022,26] hexacosa-2,5 (10),6,8,11,16,18,20,22 (26)-nonane-13,15 triazaheptacyclo[16.7.1.02,17.03,
7-(8-Quinolyl)-1,4,14- -dione -dione 7-(5-Chloro-2-hydroxyphenyl)-1,4,14- -dione 7-(o-Hydroxyphenyl)-1,4,14- (5R,7R,10R)-7-[(5-Methyl -3-isoxazolyl) methyl]-1,4,14-
BindingDB identifier BDBM6732-bio2 BDBM6732-bio1 BDBM6732-bio4 BDBM6732-bio5 BDBM6732-bio3
Volume 1 Issue 1 (2024) 120 doi: 10.36922/imo.3731