Page 35 - MSAM-1-3
P. 35
Materials Science in Additive Manufacturing Photocatalytic PA6/TiO powder for LPBF
2
A B C
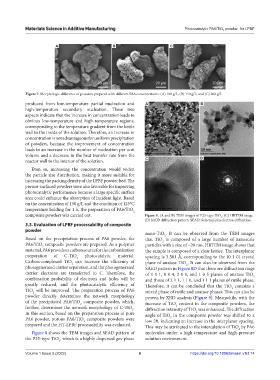
Figure 7. Morphologic difference of powders prepared with different PA6 concentrations: (A) 100 g/L, (B) 150 g/L, and (C) 200 g/L.
produced from low-temperature partial nucleation and A B
high-temperature secondary nucleation. These two
aspects indicate that the increase in concentration leads to
obvious low-temperature and high-temperature regions,
corresponding to the temperature gradient from the kettle
wall to the inside of the solution. Therefore, an increase in
concentration is not advantageous for uniform precipitation
of powders, because the improvement of concentration
leads to an increase in the number of nucleation per unit
volume and a decrease in the heat transfer rate from the
reactor wall to the interior of the solution. C D
Even so, increasing the concentration would widen
the particle size distribution, making it more suitable for
increasing the packing density of the LPBF powder bed. The
porous-surfaced powders were also favorable for improving
photocatalytic performance because a large specific surface
area could enhance the absorption of incident light. Based
on the concentration of 150 g/L and the condition of 125°C
temperature holding for 1 h, the preparation of PA6/TiO
2
composite powders was carried out. Figure 8. (A and B) TEM images of P25-type TiO . (C) HRTEM image.
2
(D) SAED diffraction pattern. SEAD: Selected area electron diffraction.
3.3. Evaluation of LPBF processability of composite
powder
nano-TiO . It can be observed from the TEM images
2
Based on the precipitation process of PA6 powder, the that TiO is composed of a large number of nanoscale
2
PA6/TiO composite powders are prepared. As a polymer particles with a size of ~20 nm. HRTEM image shows that
2
material, PA6 provides a carbon source for the carbonization the sample is composed of a clear lattice. The interplanar
preparation of C-TiO 2 photocatalytic material. spacing is 3.581 Å, corresponding to the (0 1 0) crystal
Carbon-complexed TiO can increase the efficiency of plane of anatase TiO . It can also be observed from the
2
2
photogenerated carrier separation, and the photogenerated SAED pattern in Figure 8D that there are diffraction rings
carrier electrons are transferred to C. Therefore, the of 1 0 1, 0 0 4, 2 0 0, and 1 0 5 planes of anatase TiO
2
combination probability of electrons and holes will be and those of 2 1 1, 1 1 0, and 1 1 1 planes of rutile phase.
greatly reduced, and the photocatalytic efficiency of Therefore, it can be concluded that the TiO contains a
2
TiO will be improved. The preparation process of PA6 mixed phase of rutile and anatase phases. This can also be
2
powder directly determines the network morphology proven by XRD analysis (Figure 9). Meanwhile, with the
of the precipitated PA6/TiO composite powder, which, increase of TiO content in the composite powders, the
2
2
further, determines the network morphology of C-TiO . diffraction intensity of TiO was enhanced. The diffraction
2
2
In this section, based on the preparation process of pure angle of TiO in the composite powder was shifted to a
2
PA6 powder, porous PA6/TiO composite powders were low 2θ, indicating an increase in the interplanar spacing.
2
prepared and the HT-LPBF processability was evaluated. This may be attributed to the intercalation of TiO by PA6
2
Figure 8 shows the TEM images and SEAD pattern of molecules under a high-temperature and high-pressure
the P25-type TiO , which is a highly dispersed gas-phase solution environment.
2
Volume 1 Issue 3 (2022) 6 https://doi.org/10.18063/msam.v1i3.14