Page 9 - TD-2-2
P. 9
Tumor Discovery Practice and consideration of master protocol design
A B
C
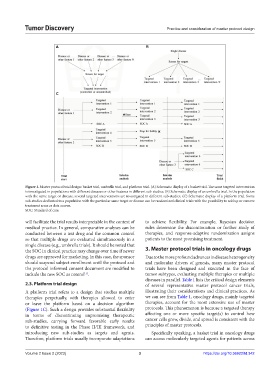
Figure 1. Master protocol trial design: basket trial, umbrella trial, and platform trial. (A) Schematic display of a basket trial. The same targeted intervention
is investigated in populations with different diseases or other features in different sub-studies. (B) Schematic display of an umbrella trial. In the population
with the same target or disease, several targeted interventions are investigated in different sub-studies. (C) Schematic display of a platform trial. Some
sub-studies dedicated to a population with the particular same target or disease can be randomized clinical trials with the possibility to add up or remove
treatment arms as data accrue.
SOC: Standard of care.
will facilitate the trial results interpretable in the context of to achieve flexibility. For example, Bayesian decision
medical practice. In general, comparative analyses can be rules determine the discontinuation or further study of
conducted between a test drug and the common control therapies, and response-adaptive randomization assigns
so that multiple drugs are evaluated simultaneously in a patients to the most promising treatment.
single disease (e.g., umbrella trials). It should be noted that
the SOC in clinical practice may change over time if newer 3. Master protocol trials in oncology drugs
drugs are approved for marketing. In this case, the sponsor Due to the more profound advances in disease heterogeneity
should suspend subject enrollment until the protocol and and molecular drivers of genesis, many master protocol
the protocol informed consent document are modified to trials have been designed and executed in the face of
include the new SOC as control . tumor subtypes, evaluating multiple therapies or multiple
[1]
diseases in parallel. Table 1 lists the critical design elements
2.3. Platform trial design of several representative master protocol cancer trials,
A platform trial refers to a design that studies multiple illustrating their considerations and clinical practices. As
therapies perpetually, with therapies allowed to enter we can see from Table 1, oncology drugs, mainly targeted
or leave the platform based on a decision algorithm therapies, account for the most extensive use of master
(Figure 1C). Such a design provides substantial flexibility protocols. This phenomenon is because a targeted therapy
in terms of discontinuing unpromising therapeutic affecting one or more specific target(s) to control how
sub-studies, carrying forward favorable early results cancer cells grow, divide, and spread is consistent with the
to definitive testing in the Phase II/III framework, and principles of master protocols.
introducing new sub-studies as targets and agents. Specifically speaking, a basket trial in oncology drugs
Therefore, platform trials usually incorporate adaptations can access molecularly targeted agents for patients across
Volume 2 Issue 2 (2023) 3 https://doi.org/10.36922/td.342