Page 10 - TD-2-2
P. 10
Tumor Discovery Practice and consideration of master protocol design
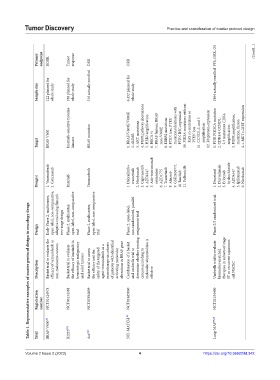
Primary endpoint BORR Tumor response ORR ORR PFS, ORR, OS (Cont’d...)
Sample size 122 planned for whole study 186 planned for whole study 216 actually enrolled 6,452 planned for whole study 1864 actually enrolled
Imatinib-sensitive tyrosine 1. BRAF/V600E/V600K 3. AKT mutations 4. FGFR pathway aberrations 5. HER2 amplification 7. BRAF fusions, BRAF 8. ERBB2 mutations 9. PTEN loss, PTEN mutations/deletions with PTEN IHC expression 10. PIK3A mutations without RAS co-mutations or PTEN loss 11. CCND1,2, and 3 amplification 12. Rb protein expression 1. PI3K PIK3CA mutation 2. CDK4/6 CCND1, 2,3 mutations CDK4 amplification
Target BRAF V600 kinases BRAF mutation 2. dMMR 6. BRCA ½ non-V600E
Drug(s) 1. Vemurafenib 2. Cetuximab Imatinib Vemurafenib 1. Dabrafenib+ trametinib 2. Nivolumab 3. Capivasertib 4. AZD4547 5. Ado-trastuzumab emtansine 6. AZD1775 7. Trametinib 8. Afatinib 9. GSK2636771 10 Taselisib 11. Palbociclib 1. Docetaxel 2. Durvalumab 3. Erlotinib hydrochloride 4. AZD4547 5. Ipilimumab 6. Nivolumab
Early Phase 2, multicenter, open-label, non-comparative, adaptive trial using Simon’s Phase 2, multicenter, open-label, non-comparative Phase 2, multicenter, open-label, non-comparative Phase 2, open-label, non-randomized, parallel Phase 2/3 randomized trial
Table 1. Representative examples of master protocol design in oncology drugs
Design two-stage design trial trial assignment trial
Basket trial to evaluate the efficacy of vemurafenib in non-melanoma cancers Basket trial to evaluate the efficacy of imatinib in hematologic malignancy Basket trial to assess safety of the targeted agent vemurafenib as a monotherapy in cohorts of patients with identified activating molecular alterations in BRAF gene Combination of a basket and umbrella trial to determine whether treating cancers according to molecular
Description and solid tumors the efficacy and the effective cell NSCLC
Registration number NCT01524978 NCT00154388 NCT02304809 NCT02465060 NCT02154490
BRAF-V600 [3] NCI-MATCH [22] Lung-MAP [9,23]
Trial B2225 [20] Ace' [21]
Volume 2 Issue 2 (2023) 4 https://doi.org/10.36922/td.342