Page 15 - TD-2-2
P. 15
Tumor Discovery Practice and consideration of master protocol design
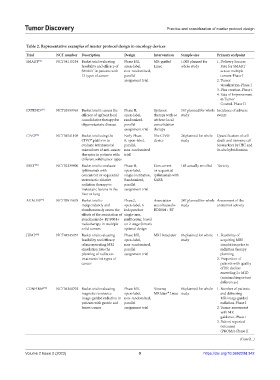
Table 2. Representative examples of master protocol design in oncology devices
Trial NCT number Description Design Intervention Sample size Primary endpoint
SMART [31] NCT04115254 Basket trial evaluating Phase I/II, MR-guided 1,000 planned for 1. Delivery Success
feasibility and efficacy of open-label, Linac whole study Rate for SMART
SMART in patients with non-randomized, across multiple
13 types of cancer parallel tumors-Phase I
assignment trial 2. Tumor
visualization-Phase I
3. Plan creation-Phase I
4. Rate of Improvement
in Tumor
Control-Phase II
EXTEND [33] NCT03599765 Basket trial to assess the Phase II, Systemic 367 planned for whole Incidence of adverse
efficacy of upfront local open-label, therapy with or study events
consolidative therapy for randomized, without local
oligometastatic disease parallel consolidation
assignment trial therapy
CIVO [34] NCT04541108 Basket trial using the Early Phase The CIVO 24 planned for whole Quantification of cell
CIVO® platform to 0, open-label, device study death and immune cell
evaluate intratumoral parallel, biomarkers by IHC and
microdoses of anti-cancer non-randomized in‑situ hybridization
therapies in patients with trial
different solid tumor types
SBRT [35] NCT02239900 Basket trial to evaluate Phase II, Concurrent 143 actually enrolled Toxicity
ipilimumab with open-label, or sequential
concurrent or sequential single-institution, ipilimumab with
stereotactic ablative Randomized, SABR
radiation therapy to parallel
metastatic lesions in the assignment trial
liver or lung
AGADIR [36] NCT03915678 Basket trial to Phase2, Association 247 planned for whole Assessment of the
independently and open-label, 6 atezolizumab+ study antitumor activity
simultaneously assess the independent BDB001+ RT
effects of the association of single-arm,
atezolizumab+ BDB001+ multicenter, based
radiotherapy in multiple on 2-stage Simon’s
solid tumors optimal design
JUMP [37] NCT04545957 Basket trial evaluating Phase I/II, MRI Simulator 86 planned for whole 1. Feasibility of
feasibility and efficacy open-label, study acquiring MRI
of incorporating MRI non-randomized, simulation prior to
simulation into the parallel radiation therapy
planning of radiation assignment trial planning
treatments in4 types of 2. Proportion of
cancer patients with quality
of life decline
exceeding 2× MID
(minimal important
differences)
CONFIRM [38] NCT04368702 Basket trial evaluating Phase I/II, Viewray 86 planned for whole 1. Number of patients
magnetic resonance open-label, MRIdian Linac study and delivering
®
image-guided radiation in non-randomized, MR-image guided
patients with gastric and parallel radiation-Phase I
breast cancer assignment trial 2. Tumor assessment
with MR
guidance-Phase I
3. Patient reported
outcomes
(PROMs)-Phase II
(Cont’d...)
Volume 2 Issue 2 (2023) 9 https://doi.org/10.36922/td.342