Page 18 - TD-2-2
P. 18
Tumor Discovery Practice and consideration of master protocol design
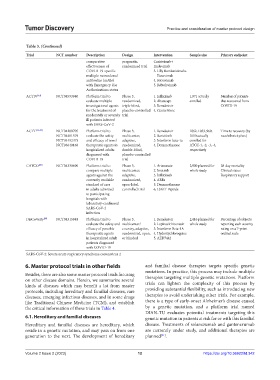
Table 3. (Continued)
Trial NCT number Description Design Intervention Sample size Primary endpoint
comparative pragmatic, Casirivimab+
effectiveness of randomized trial Imdevimab
COVID-19-specific 3. Lilly Bamlanivimab+
multiple monoclonal Etesevimab
antibodies (mAbs) 4. Sotrovimab
with Emergency Use 5. Bebtelovimab
Authorizations status
ACTIV [44] NCT04593940 Platform trial to Phase 3, 1. Infliximab 1,971 actually Number of patients
evaluate multiple randomized, 2. Abatacept enrolled that recovered from
investigational agents triple-blind, 3. Remdesivir COVID-19
for the treatment of placebo-controlled 4. Cenicriviroc
moderately or severely trial
ill patients infected
with SARS-CoV-2
ACTT [45-48] NCT04280705 Platform trial to Phase 3, 1. Remdesivir 1062, 1033, 969, Time to recovery (by
NCT04401579 evaluate the safety multicenter, 2. Baricitinib 1010 actually race/ethnicity/sex)
NCT04492475 and efficacy of novel adaptive, 3. Interferon beta-1a enrolled for
NCT04640168 therapeutic agents in randomized, 4. Dexamethasone ATCC-1, -2, -3, -4,
hospitalized adults double-blind, respectively
diagnosed with placebo-controlled
COVID-19 trial
CATCO [49] NCT04330690 Platform trial to Phase 3, 1. Artesunate 2,900 planned for 28-day mortality
compare multiple multicenter, 2. Imatinib whole study Clinical status
agents against the adaptive, 3. Infliximab Respiratory support
currently available randomized, 4. ARBs
standard of care open-label, 5. Dexamethasone
in adults admitted controlled trial 6. LSALT Peptide
to participating
hospitals with
laboratory-confirmed
SARS-CoV-2
infection
DisCoVeRy [50] NCT04315948 Platform trial to Phase 3, 1. Remdesivir 2,416 planned for Percentage of subjects
evaluate the safety and multicenter/ 2. Lopinavir/ritonavir whole study reporting each severity
efficacy of possible country, adaptive, 3. Interferon Beta-1A rating on a 7-point
therapeutic agents randomized, open, 4. Hydroxychloroquine ordinal scale
in hospitalized adult or blinded 5. AZD7442
patients diagnosed
with COVID-19
SARS-CoV-2: Severe acute respiratory syndrome coronavirus 2
6. Master protocol trials in other fields and familial disease therapies targets specific genetic
mutations. In practice, this process may include multiple
Besides, there are also some master protocol trials focusing therapies targeting multiple genetic mutations. Platform
on other disease domains. Herein, we summarize several trials can lighten the complexity of this process by
kinds of diseases which may benefit a lot from master
protocols, including hereditary and familial diseases, rare providing substantial flexibility, such as introducing new
diseases, emerging infectious diseases, and in some drugs therapies to avoid undertaking other trials. For example,
like Traditional Chinese Medicine (TCM), and establish there is a type of early-onset Alzheimer’s disease caused
the critical information of these trials in Table 4. by a genetic mutation, and a platform trial named
DIAN-TU evaluates potential treatments targeting this
6.1. Hereditary and familial diseases genetic mutation in patients at risk for or with this familial
Hereditary and familial diseases are hereditary, which disease. Treatments of solanezumab and gantenerumab
reside in a genetic mutation, and may pass on from one are currently under study, and additional therapies are
[51]
generation to the next. The development of hereditary planned .
Volume 2 Issue 2 (2023) 12 https://doi.org/10.36922/td.342