Page 17 - TD-2-2
P. 17
Tumor Discovery Practice and consideration of master protocol design
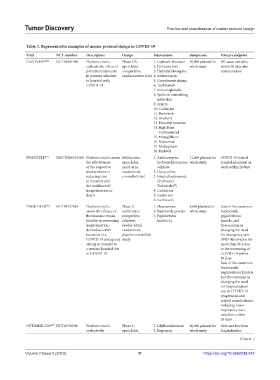
Table 3. Representative examples of master protocol design in COVID‑19
Trial NCT number Description Design Intervention Sample size Primary endpoint
RECOVERY [40] NCT04381936 Platform trial to Phase 2/3, 1. Lopinavir-Ritonavir 50,000 planned for All-cause mortality
evaluate the effects of open-label, 2. Corticosteroid whole study within 28 days after
potential treatments comparative, 3. Hydroxychloroquine randomization
in patients admitted randomization trial 4. Azithromycin
to hospital with 5. Convalescent plasma
COVID-19 6. Tocilizumab
7. Immunoglobulin
8. Synthetic neutralising
antibodies
9. Aspirin
10. Colchicine
11. Baricitinib
12. Anakinra
13. Dimethyl fumarate
14. High Dose
Corticosteroid
15. Empagliflozin
16. Sotrovimab
17. Molnupiravir
18. Paxlovid
PRINCIPLE [41] ISRCTN86534580 Platform trial to assess Multicenter, 1. Azithromycin, 12,000 planned for COVID-19-related
the effectiveness open-label, hydroxychloroquine whole study hospital admission or
of the respective multi-arm, sulphate death within 28 days
interventions in randomized, 2. Doxycycline
reducing time controlled trial 3. Inhaled budesonide
to recovery and (Pulmicort
the incidence of Turbohaler )
®
hospitalization or 4. Colchicine
death. 5. Favipiravir
6. Ivermectin
TOGETHER [42] NCT04727424 Platform trial to Phase 3, 1. Fluvoxamine 4,669 planned for Rate of fluvoxamine+
assess the efficacy of multicenter, 2. Budesonide powder whole study budesonide,
fluvoxamine versus prospective, 3. Peginterferon peginterferon
placebo in preventing adaptive, lambda-1a lambda, and
hospitalization double-blind, fluvoxamine in
defined as either randomized, changing the need
retention in a placebo-controlled for emergency care
COVID-19 emergency study AND observation for
setting or transfer to more than 06 h due
a tertiary hospital due to the worsening of
to COVID-19 COVID-19 within
28 days
Rate of fluvoxamine+
budesonide,
peginterferon lambda
and fluvoxamine in
changing the need
for hospitalization
due to COVID-19
progression and
related complications,
including lower
respiratory tract
infection within
28 days
OPTIMISE-C19 [43] NCT04790786 Platform trial to Phase 4, 1. LillyBamlanivimab 60,000 planned for Alive and free from
evaluate the open-label, 2. Regeneron whole study hospitalization
(Cont’d...)
Volume 2 Issue 2 (2023) 11 https://doi.org/10.36922/td.342