Page 19 - TD-2-2
P. 19
Tumor Discovery Practice and consideration of master protocol design
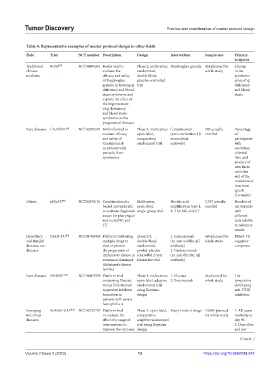
Table 4. Representative examples of master protocol design in other fields
Field Trial NCT number Description Design Intervention Sample size Primary
endpoint
Traditional BOSS [57] NCT04408261 Basket trial to Phase 2, multicenter, Buqitongluo granule 432 planned for Change
chinese evaluate the randomized, whole study in the
medicine efficacy and safety double-blind, syndrome
of Buqitongluo placebo-controlled score of qi
granule in treating qi trial deficiency
deficiency and blood and blood
stasis syndrome and stasis
explore the effect of
the improvement
of qi deficiency
and blood stasis
syndrome on the
prognosis of diseases
Rare diseases CLUSTER [59] NCT02059291 Umbrella trial to Phase 3, multicenter, Canakinumab 203 actually Percentage
evaluate efficacy open-label, (anti-interleukin-1β enrolled of
and safety of comparative, monoclonal participants
Canakinumab randomized trial antibody) with
in patients with resolution
periodic fever of initial
syndromes flare and
absence of
new flares
up to the
end of the
randomized
treatment
epoch
(16 weeks)
Others pNAAT [60] NCT02870101 Combination of a Multicenter, Nucleic acid 2,767 actually Number of
basket and umbrella open-label, amplification tests 1, enrolled participants
to evaluate diagnostic single-group trial 2, 3 for NG and CT with
assays for pharyngeal different
and rectal NG and tests relative
CT to reference
results
Hereditary DIAN-TU [51] NCT01760005 Platform trial testing phase 2/3, 1. Solanezumab 490 planned for DIAN-TU
and familial multiple drugs to double-blind, (an anti-soluble Aβ whole study cognitive
diseases, are slow or prevent randomized, antibody) composite
diseases the progression of pooled-placebo 2. Gantenerumab
Alzheimer’s disease in controlled 2-year (an anti-fibrillar Aβ
autosomal dominant biomarker trial antibody)
Alzheimer’s disease
families
Rare diseases INHIBIT [53] NCT04303559 Platform trial Phase 3, multicenter, 1. Eloctate 66 planned for The
comparing Eloctate open-label, adaptive 2. Emicizumab whole study proportion
versus Emicizumab randomized trial developing
to prevent inhibitor using Bayesian anti-FVIII
formation in design inhibitors
patients with severe
hemophilia A
Emerging REMAP-CAP [54] NCT02735707 Platform trial Phase 3, open-label, Many kinds of drugs 10,000 planned 1. All-cause
infectious to evaluate the comparative, for whole study mortality at
diseases effect of a range of adaptive randomized day 90
interventions to trial using Bayesian 2. Days alive
improve the outcome design and not
(Cont’d...)
Volume 2 Issue 2 (2023) 13 https://doi.org/10.36922/td.342