Page 52 - AN-3-3
P. 52
Advanced Neurology HS-proteoglycans and brain function
(32kDa) lacks the KPI-3 domain. TFPI functions as an the HGF activator, thereby inhibiting the conversion of
anticoagulant protein that blocks the initiation of blood pro-HGF into its active form.
coagulation by inhibiting the tissue factor/activated
factor VIII (TF–fVIIa) complex and thrombin activation. 5.4. Amyloid precursor protein (APP)
A study has demonstrated that Tfpi mice die during The APP contains a KPI domain (protease nexin 2), which
-/-
embryonic development, displaying stunted brain growth, acts as a highly specific substrate for mesotrypsin, atypical
delayed meningeal development, and severe vascular human trypsin implicated in a number of neurodegenerative
34
pathology. The generation of thrombin is associated with disorders (Figure 2A). Neurotrypsin also cleaves agrin
36
hemorrhagic stroke and is central to the pathogenesis of locally at the synapse. The KPI domain in APP inhibits pro-
acute ischemic stroke. TFPI-2 exhibits neuroprotective neuropeptide processing, and the accumulation of APP
properties, inhibits human glioblastoma formation, and in Aβ plaques may hinder their clearance from tissues,
inhibits a range of proteases, including plasmin, trypsin, thereby prolonging their neurotoxic effects in AD and
chymotrypsin, cathepsin G, plasma kallikrein, FVIIa, Fxa. PD. The accumulation of Aβ arises from the processing of
In addition, TFPI-2 inhibits fibrinolysis, thereby regulating APP-1 by β- and γ-secretases in the brain, an early toxic
pericellular proteases that are implicated in cancer cell event in the pathogenesis of AD that leads to the formation
invasion, vascular inflammation, and atherosclerosis. of disruptive intracellular neurofibrillary tangles and
TFPI2 has been proposed as a tumor repressor gene in extracellular Aβ senile plaques. APP is also cleaved by
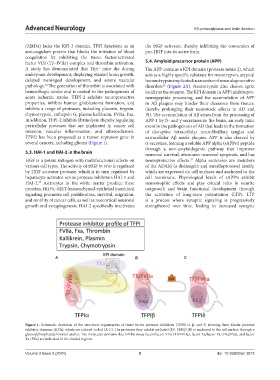
several cancers, including glioma (Figure 1). α-secretase, forming a soluble APP alpha (sAPPα) peptide
through a non-amyloidogenic pathway that improves
5.3. HAI-1 and HAI-2 in the brain neuronal survival, attenuates neuronal apoptosis, and has
HGF is a potent mitogen with multifunctional effects on neuroprotective effects. Alpha secretases are members
37
various cell types. The activity of HGF in vivo is regulated of the ADAM (a disintegrin and metalloprotease) family,
by HGF activator protease, which is in turn regulated by which are expressed on cell surfaces and anchored to the
hepatocyte activator serine protease inhibitors HAI-1 and cell membrane. Physiological levels of sAPPα exhibit
HAI-2. Astrocytes in the white matter produce these neurotrophic effects and play critical roles in neurite
35
proteins. HGF/c-MET (mesenchymal-epithelial transition) outgrowth and brain functional development through
signaling promotes cell proliferation, survival, migration, the activation of long-term potentiation (LTP). LTP
and motility of cancer cells, as well as neocortical neuronal is a process where synaptic signaling is progressively
growth and synaptogenesis. HAI-2 specifically inactivates strengthened over time, leading to increased synaptic
A B C
Figure 1. Schematic depiction of the structural organization of tissue factor protease inhibitors (TFPI) α, β, and δ, showing their Kunitz protease
inhibitor domains (KPIs), which are colored-coded (A-C). The proteases they inhibit are listed (D). TFPIβ (B) is anchored to the cell surface through a
glycosylphosphatidylinositol anchor. The molecular domains that inhibit tissue factor/factor VIIa (TF/FVIIa), factor Xa/factor Va (FXa/FVa), and factor
Xa (FXa) are indicated in the shaded regions.
Volume 3 Issue 3 (2024) 5 doi: 10.36922/an.3812