Page 53 - AN-3-3
P. 53
Advanced Neurology HS-proteoglycans and brain function
A
B
C
D
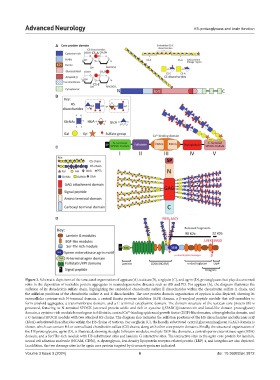
Figure 2. Schematic depictions of the structural organization of appican (A), testican (B), serglycin (C), and agrin (D), proteoglycans that play documented
roles in the deposition of insoluble protein aggregates in neurodegenerative diseases such as AD and PD. For appican (A), the diagram illustrates the
sulfation of its chondroitin sulfate chain, highlighting the embedded chondroitin sulfate E disaccharides within the chondroitin sulfate A chain, and
the sulfation positions of the chondroitin sulfate A and E disaccharides. The core protein domain organization of appican is also depicted, showing its
extracellular cysteine-rich N-terminal domain, a central Kunitz protease inhibitor (KPI) domain, a β-amyloid peptide module that self-assembles to
form amyloid aggregates, a transmembrane domain, and a C-terminal cytoplasmic domain. The domain structure of the testican core protein (B) is
presented, featuring its N-terminal SPOCK (secreted protein acidic and rich in cysteine ([SPARC)]/osteonectin and kazal-like domain proteoglycan)
domain, a cysteine-rich module homologous to follistatin, central Ca -binding epidermal growth factor (EGF)-like domains, a thyroglobulin domain, and
2+
a C-terminal SPOCK module with two attached HS chains. The diagram also indicates the sulfation positions of the HS disaccharides and iduronic acid
(IdoA)-substituted disaccharides within the HS chains of testican. For serglycin (C), the heavily substituted central glycosaminoglycan (GAG) domain is
shown, which can contain HS or oversulfated chondroitin sulfate (CS) chains, along with other core protein domains. Finally, the structural organization of
the HS proteoglycans, agrin (D), is illustrated, showing its eight follistatin modules, multiple EGF-like domains, a central sperm-enterokinase-agrin)SEA)
domain, and a Ser/Thr-rich module with HS substitution sites and laminin G interactive sites. The interactive sites in the agrin core protein for laminin,
neural cell adhesion molecule (NCAM, CD56), α-dystroglycan, low-density lipoprotein receptor-related protein (LRP) 4, and integrins are also depicted.
In addition, the two cleavage sites in the agrin core protein targeted by α-neurotrypsin are indicated.
Volume 3 Issue 3 (2024) 6 doi: 10.36922/an.3812