Page 66 - AN-3-3
P. 66
Advanced Neurology mTOR inhibition in epilepsy
cognition. With the emergence and rapid advancement 2. mTOR signaling pathway
5
of genomic technologies, our understanding of epilepsy
genetics has expanded, revealing critical pathways involved The mTOR signaling pathway is crucial for regulating
in epileptogenesis beyond the final common mechanisms cellular processes such as growth, proliferation, apoptosis,
of ion channels. This expanded understanding has opened autophagy, metabolism, and cytoskeletal organization, as
new avenues for the development of precision medicine. well as brain-specific functions such as synaptic plasticity,
6,7
12-14
Both dysregulated mammalian or mechanistic targets of neurogenesis, and dendritic-axonal morphology.
the rapamycin (mTOR) signaling pathway and heightened The mTOR protein is a serine-threonine protein kinase
neuroinflammation have been implicated in epileptogenesis belonging to the phosphatidylinositol-3-kinase (PI3K)-
by causing hyperexcitability, altered synaptic transmission, related protein kinase family. It consists of several domains,
and increased seizure susceptibility. Inhibition of these such as FAT (FK506-binding proteins [FKBP]-rapamycin-
8
processes could be a promising therapeutic option with associated protein [FRAP]; ataxia-telangiectasia mutated
potential antiepileptogenic or disease-modifying effects. 8 [ATM]; transformation/transcription-domain-associated
protein; and TRRAP), FKBP 12-rapamycin-binding (FRB),
Over the past two decades, there has been a drastic FATC (FRAP, ATM, and TRRAP C-terminal), kinase
increase in research on the mTOR signaling pathway domains, and HEAT (Huntington, elongation factor 3,
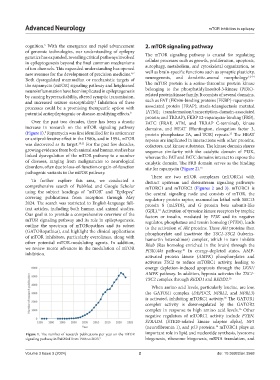
(Figure 1). Rapamycin was first identified for its anticancer protein phosphatase 2A, and TOR) repeats. The HEAT
9
13
or antiproliferative effect in the 1980s, and in 1994, mTOR repeats are implicated in interactions with other proteins,
was discovered as its target. 10,11 For the past few decades, cofactors, and kinase substrates. The kinase domain shares
growing evidence from both animal and human studies has sequence similarity with the catalytic domain of PI3K,
linked dysregulation of the mTOR pathway to a number whereas the FAT and FATC domains interact to expose the
of diseases, ranging from malignancies to neurological catalytic domain. The FRB domain serves as the binding
disorders, often due to loss-of-function or gain-of-function site for rapamycin (Figure 2). 15
pathogenic variants in the mTOR pathway.
There are two mTOR complexes (mTORCs) with
To further explore this area, we conducted a distinct upstream and downstream signaling pathways:
comprehensive search of PubMed and Google Scholar mTORC1 and mTORC2 (Figures 2 and 3). mTORC1 is
using the subject headings of “mTOR” and “Epilepsy,” the central signaling node and consists of mTOR, the
covering publications from inception through May regulatory protein raptor, mammalian lethal with SEC13
2024. The search was restricted to English-language full- protein 8 (mLST8), and G protein beta subunit-like
text articles, including both human and animal studies. (GβL). Activation of tyrosine kinase receptors by trophic
16
Our goal is to provide a comprehensive overview of the factors or insulin, mediated by PI3K and its negative
mTOR signaling pathway and its role in epileptogenesis, regulator, phosphatase and tensin homolog (PTEN), leads
outline the spectrum of mTORopathies and its subset to the activation of Akt proteins. These Akt proteins then
(GATORopathies), and highlight the clinical applications phosphorylate and inactivate the TSC1-TSC2 (tuberin-
of mTOR inhibitors, particularly everolimus, along with hamartin heterodimer) complex, which in turn inhibits
other potential mTOR-modulating agents. In addition, Rheb (Ras homolog enriched in the brain) through the
we review recent advances in the modulation of mTOR PI3K/Akt pathway. In energy-depleted states, AMP-
16
inhibition. activated protein kinase (AMPK) phosphorylates and
activates TSC2 to reduce mTORC1 activity, leading to
energy depletion-induced apoptosis through the LKB1/
AMPK pathway. In addition, hypoxia activates the TSC1-
TSC2 complex through REDD1 and REDD2. 16
When amino acid levels, particularly leucine, are low,
the GATOR1 complex (DEPDC5, NPRL2, and NPRL3)
is activated, inhibiting mTORC1 activity. The GATOR1
16
complex activity is downregulated by the GATOR2
complex in response to high amino acid levels. Other
16
negative regulators of mTORC1 activity include PTEN,
STRADA (STE20-related kinase adaptor alpha), NF1
(neurofibromin 1), and p53 proteins. mTORC1 plays an
16
Figure 1. The number of research publications per year on the mTOR important role in lipid and nucleotide synthesis, lysosome
signaling pathway in PubMed from 1986 to 2023. 9 biogenesis, ribosome biogenesis, mRNA translation, and
Volume 3 Issue 3 (2024) 2 doi: 10.36922/an.3568