Page 71 - AN-3-3
P. 71
Advanced Neurology mTOR inhibition in epilepsy
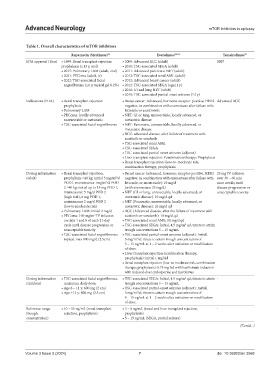
Table 1. Overall characteristics of mTOR inhibitors
Rapamycin (Sirolimus) 51 Everolimus 52‑54 Temsirolimus 55
FDA approval (Year) • 1999: Renal transplant rejection • 2009: Advanced RCC (adult) 2007
prophylaxis (≥13 y, oral) • 2010: TSC‑associated SEGA (adult)
• 2015: Pulmonary LAM (adult, oral) • 2011: Advanced pancreatic NET (adult)
• 2021: PEComa (adult, iv) • 2012: TSC‑associated renal AML (adult)
• 2022: TSC‑associated facial • 2012: Advanced breast cancer (adult)
angiofibroma (≥6 y, topical gel 0.2%) • 2012: TSC‑associated SEGA (age≥1 y)
• 2016: GI and lung NET (adult)
• 2018: TSC‑associated partial‑onset seizures (≥2 y)
Indications (FDA) • Renal transplant rejection • Breast cancer: Advanced, hormone receptor‑positive, HER2 Advanced RCC
prophylaxis negative, in combination with exemestane after failure with
• Pulmonary LAM letrozole or anastrozole
• PEComa: locally advanced • NET: GI or lung, unresectable, locally advanced, or
unresectable or metastatic metastatic disease
• TSC‑associated facial angiofibroma • NET: Pancreatic, unresectable, locally advanced, or
metastatic disease
• RCC: advanced disease, after failure of treatment with
sunitinib or sorafenib
• TSC‑associated renal AML
• TSC‑associated SEGA
• TSC‑associated partial‑onset seizures (adjunct)
• Liver transplant rejection: Combination therapy; Prophylaxis
• Renal transplant rejection: Low‑to‑moderate risk,
combination therapy; prophylaxis
Dosing information • Renal transplant rejection, • Breast cancer (advanced, hormone receptor‑positive, HER2 25 mg IV infusion
(adult) prophylaxis <40 kg: initial 3 mg/m /d negative, in combination with exemestane after failure with over 30 – 60 min
2
POD1, maintenance 1mg/m /d POD letrozole, or anastrozole): 10 mg/d once weekly until
2
2 ≥40 kg: initial up to 15 mg POD 1, (with exemestane 25 mg/d) disease progression or
maintenance 5 mg/d POD 2 • NET (GI or lung, unresectable, locally advanced, or unacceptable toxicity
(high risk), 6 mg POD 1, metastatic disease): 10 mg/d qd
maintenance 2 mg/d POD 2 • NET (Pancreatic, unresectable, locally advanced, or
(low to moderate risk) metastatic disease): 10 mg/d qd
• Pulmonary LAM: initial 2 mg/d • RCC (Advanced disease, after the failure of treatment with
• PEComa: 100 mg/m IV infusion sunitinib or sorafenib): 10 mg/d qd
2
on days 1 and 8 of each 21-day • TSC‑associated renal AML: 10 mg/d qd
cycle until disease progression or • TSC‑associated SEGA: Initial, 4.5 mg/m qd, titrate to attain
2
unacceptable toxicity trough concentrations 5 – 15 ng/mL
• TSC‑associated facial angiofibroma: • TSC‑associated partial‑onset seizures (adjunct): Initial,
topical, max 800 mg/d (2.5cm) 5 mg/m /d; titrate to attain trough concentration of
2
5 – 15 ng/mL at 1 – 2 weeks after initiation or modification
of dose.
• Liver transplant rejection (combination therapy;
prophylaxis): initial 1 mg bid
• Renal transplant rejection (low‑to‑moderate risk, combination
therapy; prophylaxis): 0.75 mg bid with basiliximab induction
with reduced-dose cyclosporine and tacrolimus
Dosing information • TSC‑associated facial angiofibroma, • TSC‑associated SEGA: Initial, 4.5 mg/m qd, titrate to attain -
2
(children) maximum daily dose: trough concentrations 5 – 15 ng/mL
• Age 6 – 11 y: 600 mg (2 cm) • TSC‑associated partial‑onset seizures (adjunct): Initial,
• Age ≥12 y: 800 mg (2.5 cm) 5 mg/m /d; titrate to attain trough concentration of
2
5 – 15 ng/mL at 1 – 2 weeks after initiation or modification
of dose.
Reference range • 10 – 15 ng/mL (renal transplant • 3 – 8 ng/mL (renal and liver transplant rejection, -
(trough rejection, prophylaxis) prophylaxis)
concentration) • 5 – 15 ng/mL (SEGA, partial seizure)
(Contd...)
Volume 3 Issue 3 (2024) 7 doi: 10.36922/an.3568