Page 73 - AN-3-3
P. 73
Advanced Neurology mTOR inhibition in epilepsy
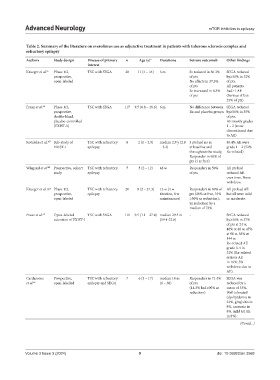
Table 2. Summary of the literature on everolimus use as adjunctive treatment in patients with tuberous sclerosis complex and
refractory epilepsy
Authors Study design Disease of primary n Age (y) a Durationa Seizure outcomeb Other findings
interest
Krueger et al. 65 Phase 1/2, TSC with SEGA 28 11 (3 – 34) 6 m Sz reduced in 56.3% SEGA reduced
prospective, of pts by≥50% in 32%
open-labeled No effects in 37.5% of pts.
of pts All patients
Sz increased in 6.3% had≥1 AE
of pts (Serious AE in
25% of pts)
Franz et al. 66 Phase 1/2, TSC with SEGA 117 9.5 (0.8 – 26.6) 6 m No difference between SEGA reduced
prospective, Rx and placebo groups by≥50% in 35%
double-blind, of pts.
placebo-controlled AE mostly grades
(EXIST-1) 1 – 2 (none
discontinued due
to AE)
Kotulska et al. 107 Sub-study of TSC with refractory 8 2 (1 – 2.9) median 2.9 y (2.8 3 pts had no sz 89.4% AE were
EXIST-1 epilepsy – 3.2) at baseline and grade 1 – 2 (52%
throughout the study; Rx-related)
Responder in 60% of
pts (1 sz free)
Wiegand et al 108 Prospective, cohort TSC with refractory 7 5 (2 – 12) 48 w Responders in 50% All pts had
study epilepsy of pts reduced AE
over time. None
withdrew.
Krueger et al. 67 Phase 1/2, TSC with refractory 20 8 (2 – 21.3) 12 w (4 w Responders in 60% of All pts had AE
prospective, epilepsy titration, 8 w pts (20% sz free, 35% but all were mild
open-labeled maintenance) ≥90% sz reduction); or moderate.
Sz reduction by a
median of 73%
Franz et al. 68 Open-labeled TSC with SEGA 111 9.5 (1.1 – 27.4) median 29.3 m - SEGA reduced
extension of EXIST-1 (19.4-33.8) by≥50% in 37%
of pts at 24 w,
46% at 48 w, 47%
at 96 w, 38% at
144 w.
Rx-related AE
grade 3-4 in
32% (Rx-related
serious AE
in 16%; 5%
withdrew due to
AE).
Cardamone Prospective, TSC with refractory 7 6 (3 – 17) median 18 m Responders in 71.4% SEGA was
et al. 63 open-labelled epilepsy and SEGA (6 – 38) of pts reduced by a
(14.3% had≥90% sz mean of 33%.
reduction) Well tolerated
(dyslipidemia in
23%, gingivitis in
8%, anorexia in
8%, mild GI AE
in 8%)
(Contd...)
Volume 3 Issue 3 (2024) 9 doi: 10.36922/an.3568