Page 77 - AN-3-3
P. 77
Advanced Neurology mTOR inhibition in epilepsy
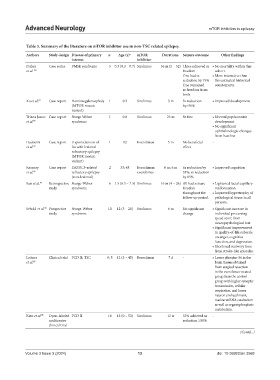
Table 3. Summary of the literature on mTOR inhibitor use in non‑TSC related epilepsy.
Authors Study design Disease of primary n Age (y) a mTOR Durationa Seizure outcome Other findings
interest inhibitor
Parker Case series PMSE syndrome 5 0.3 (0.3 – 0.7) Sirolimus 36 m (5 – 52) Three achieved sz • No mortality within this
et al. 80 freedom cohort.
One had sz • More interactive than
reduction by 75% the untreated historical
One remained counterparts.
sz freedom from
birth
Xu et al. 81 Case report Hemimegalencephaly 1 0.3 Sirolimus 3 m Sz reduction • Improved development
(MTOR mosaic by>50%
variant)
Triana Junco Case report Sturge Weber 1 0.8 Sirolimus 23 m Sz free • Normal psychomotor
et al. 92 syndrome development
• No significant
ophthalmologic changes
from baseline
Hadouiri Case report Hypomelanosis of 1 12 Everolimus 5 m No beneficial -
et al. 82 Ito with lesional effect
refractory epilepsy
(MTOR mosaic
variant)
Kearney Case report DEPDC5-related 2 33; 48 Everolimus; 6 m; 6 m Sz reduction by • Improved cognition
et al. 89 refractory epilepsy everolimus 33%; sz reduction
(non-lesional) by 85%
Sun et al. 93 Retrospective Sturge-Weber 6 1.5 (0.5 – 7.5) Sirolimus 16 m (4 – 26) All had seizure • Lightened facial capillary
study syndrome freedom malformation
throughout the • Improved hypertrophy of
follow-up period. pathological tissue in all
patients.
Sebold et al. 94 Prospective Sturge-Weber 10 12 (5 – 20) Sirolimus 6 m No significant • Significant increase in
study syndrome change individual processing
speed score from
neuropsychological test.
• Significant improvement
in quality-of-life subscale
on anger, cognitive
function, and depression.
• Shortened recovery time
from stroke-like episodes.
Leitner Clinical trial FCD II; TSC 9; 5 12 (3 – 45) Everolimus 7 d - • Lower phospho‑S6 in the
et al. 83 brain tissues obtained
from surgical resection
in the everolimus-treated
group than the control
group with higher synaptic
transmission, cellular
respiration, and lower
neuron ensheathment,
nuclear mRNA catabolism
as well as organophosphate
metabolism.
Kato et al. 85 Open-labeled FCD II 16 13 (0 – 53) Sirolimus 12 w 33% achieved sz -
multicenter reduction ≥50%
clinical trial
(Contd...)
Volume 3 Issue 3 (2024) 13 doi: 10.36922/an.3568