Page 85 - AN-4-4
P. 85
Advanced Neurology Cytokine response to EV therapy in SCI
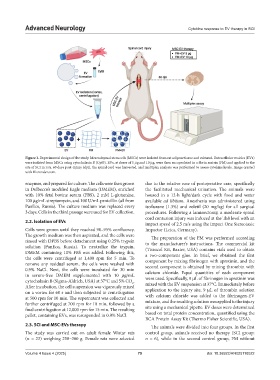
Figure 1. Experimental design of the study. Mesenchymal stem cells (MSCs) were isolated from rat adipose tissue and cultured. Extracellular vesicles (EVs)
were isolated from MSCs using cytochalasin B (CytB). EVs, at doses of 5 µg and 10 µg, were then encapsulated in a fibrin matrix (FM) and applied to the
site of SCI in rats. 60 days post-injury (dpi), the spinal cord was harvested, and multiplex analysis was performed to assess cytokine levels. Image created
with Biorender.com.
enzymes, and prepared for culture. The cells were then grown due to the relative ease of postoperative care, specifically
in Dulbecco’s modified Eagle medium (DMEM), enriched the facilitated mechanical urination. The animals were
with 10% fetal bovine serum (FBS), 2 mM L-glutamine, housed in a 12-h light/dark cycle with food and water
100 µg/mL streptomycin, and 100 U/mL penicillin (all from available ad libitum. Anesthesia was administered using
PanEco, Russia). The culture medium was replaced every isoflurane (1.3%) and zoletil (20 mg/kg) for all surgical
3 days. Cells in the third passage were used for EV collection. procedures. Following a laminectomy, a moderate spinal
cord contusion injury was induced at the Th8 level with an
2.2. Isolation of EVs impact speed of 2.5 m/s using the Impact One Stereotaxic
Cells were grown until they reached 90–95% confluency. Impactor (Leica, Germany).
The growth medium was then aspirated, and the cells were The preparation of the FM was performed according
rinsed with DPBS before detachment using 0.25% trypsin to the manufacturer’s instructions. The commercial kit
solution (PanEco, Russia). To neutralize the trypsin, (Tissucol Kit, Baxter, USA) contains vials used to obtain
DMEM containing 10% FBS was added. Following this, a two-component glue. In brief, we obtained the first
the cells were centrifuged at 1,400 rpm for 5 min. To component by mixing fibrinogen with aprotinin, and the
remove any residual serum, the cells were washed with second component is obtained by mixing thrombin with
0.9% NaCl. Next, the cells were incubated for 30 min
in serum-free DMEM supplemented with 10 µg/mL calcium chloride. Equal quantities of each component
cytochalasin B (Sigma-Aldrich, USA) at 37°C and 5% CO . were used. Specifically, 9 µL of fibrinogen in aprotinin was
mixed with the EV suspension at 37°C. Immediately before
2
After incubation, the cell suspension was vigorously mixed application to the injury site, 9 µL of thrombin solution
on a vortex for 60 s and then subjected to centrifugation
at 500 rpm for 10 min. The supernatant was collected and with calcium chloride was added to the fibrinogen-EV
further centrifuged at 700 rpm for 10 min, followed by a mixture, and the resulting solution was applied to the injury
final centrifugation at 12,000 rpm for 15 min. The resulting site using a mechanical pipette. EV doses were determined
pellet, containing EVs, was resuspended in 0.9% NaCl. based on total protein concentration, quantified using the
BCA Protein Assay Kit (Thermo Fisher Scientific, USA).
2.3. SCI and MSC-EVs therapy The animals were divided into four groups. In the first
The study was carried out on adult female Wistar rats control group, animals received no therapy (SCI group:
(n = 23) weighing 250–300 g. Female rats were selected n = 6), while in the second control group, FM without
Volume 4 Issue 4 (2025) 79 doi: 10.36922/AN025110022