Page 45 - ARNM-1-1
P. 45
Advances in Radiotherapy
& Nuclear Medicine CCRT plus nimotuzumab for cervical cancer
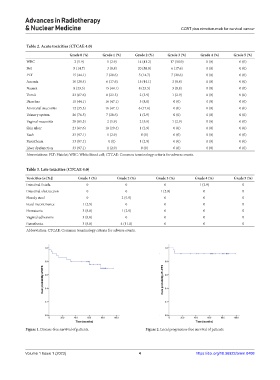
Table 2. Acute toxicities (CTCAE 4.0)
Grade 0 (%) Grade 1 (%) Grade 2 (%) Grade 3 (%) Grade 4 (%) Grade 5 (%)
WBC 2 (5.9) 1 (2.9) 14 (41.2) 17 (50.0) 0 (0) 0 (0)
Net 5 (14.7) 3 (8.8) 20 (58.8) 6 (17.6) 0 (0) 0 (0)
PLT 15 (44.1) 7 (20.6) 5 (14.7) 7 (20.6) 0 (0) 0 (0)
Anemia 10 (29.4) 6 (17.6) 15 (44.1) 3 (8.8) 0 (0) 0 (0)
Nausea 8 (23.5) 15 (44.1) 8 (23.5) 3 (8.8) 0 (0) 0 (0)
Vomit 23 (67.6) 8 (23.5) 2 (5.9) 1 (2.9) 0 (0) 0 (0)
Diarrhea 15 (44.1) 16 (47.1) 3 (8.8) 0 (0) 0 (0) 0 (0)
Anorectal mucositis 12 (35.3) 16 (47.1) 6 (17.6) 0 (0) 0 (0) 0 (0)
Urinary system 26 (76.5) 7 (20.6) 1 (2.9) 0 (0) 0 (0) 0 (0)
Vaginal mucositis 29 (85.3) 2 (5.9) 2 (5.9) 1 (2.9) 0 (0) 0 (0)
Skin ulcer 23 (67.6) 10 (29.4) 1 (2.9) 0 (0) 0 (0) 0 (0)
Rash 33 (97.1) 1 (2.9) 0 (0) 0 (0) 0 (0) 0 (0)
Paresthesia 33 (97.1) 0 (0) 1 (2.9) 0 (0) 0 (0) 0 (0)
Liver dysfunction 33 (97.1) 1 (2.9) 0 (0) 0 (0) 0 (0) 0 (0)
Abrreviations: PLT: Platelet; WBC: White blood cell; CTCAE: Common terminology criteria for adverse events.
Table 3. Late toxicities (CTCAE 4.0)
Toxicities (n [%]) Grade 1 (%) Grade 2 (%) Grade 3 (%) Grade 4 (%) Grade 5 (%)
Intestinal fistula 0 0 0 1 (2.9) 0
Intestinal obstruction 0 0 1 (2.9) 0 0
Bloody stool 0 2 (5.9) 0 0 0
Fecal incontinence 1 (2.9) 0 0 0 0
Hematuria 3 (8.8) 1 (2.9) 0 0 0
Vaginal adhesions 3 (8.8) 0 0 0 0
Paresthesia 3 (8.8) 4 (11.8) 0 0 0
Abbreviation: CTCAE: Common terminology criteria for adverse events.
Figure 1. Disease-free survival of patients. Figure 2. Local progression-free survival of patients.
Volume 1 Issue 1 (2023) 4 https://doi.org/10.36922/arnm.0408