Page 95 - EER-1-1
P. 95
Explora: Environment
and Resource Accelerated carbonation for heavy metal inhibition in incineration bottom ash
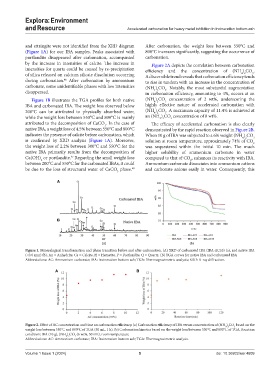
and ettringite were not identified from the XRD diagram After carbonation, the weight loss between 550°C and
(Figure 1A) for our IBA samples. Peaks associated with 800°C increases significantly, suggesting the occurrence of
portlandite disappeared after carbonation, accompanied carbonation.
by the increase in intensities of calcite. The increase in Figure 2A depicts the correlation between carbonation
intensities for quartz could be caused by re-precipitation efficiency and the concentration of (NH ) CO .
4 2
3
of silica released on calcium silicate dissolution occurring A discernible trend reveals that carbonation efficiency tends
during carbonation. After carbonation by ammonium to rise in tandem with an increase in the concentration of
46
carbonate, some unidentifiable phases with low intensities (NH ) CO . Notably, the most substantial augmentation
4 2
3
disapperaed. in carbonation efficiency, amounting to 4%, occurs at an
Figure 1B illustrates the TGA profiles for both native (NH ) CO concentration of 2 wt%, underscoring the
4 2
3
IBA and carbonated IBA. The weight loss observed below highly effective nature of accelerated carbonation with
200°C can be attributed to physically absorbed water, (NH ) CO . A maximum capacity of 11.4% is achieved at
4 2
3
while the weight loss between 550°C and 800°C is mainly an (NH ) CO concentration of 8 wt%.
3
4 2
attributed to the decomposition of CaCO . In the case of The efficacy of accelerated carbonation is also clearly
3
native IBA, a weight loss of 4.5% between 550°C and 800°C demonstrated by the rapid reaction observed in Figure 2B.
indicates the presence of calcite before carbonation, which When 10 g of IBA was subjected to a 6% weight (NH ) CO
4 2
3
is confirmed by XRD analysis (Figure 1A). Moreover, solution at room temperature, approximately 74% of CO
2
the weight loss of 2.2% between 300°C and 550°C for the was sequestered within the initial 10 min. The much
native IBA primarily results from the decomposition of higher solubility of ammonium carbonate in water
Ca(OH) or portlandite. Regarding the small weight loss compared to that of CO , enhances its reactivity with IBA.
47
2
2
between 200°C and 550°C for the carbonated IBAs, it could Ammonium carbonate dissociates into ammonium cations
be due to the loss of structural water of CaCO phase. and carbonate anions easily in water. Consequently, this
48
3
A
B
Figure 1. Mineralogical transformation and phase transition before and after carbonation. (A) XRD of carbonated IBA (IBA-AC10) (a), and native IBA
(<0.6 mm) (b). An = Anhydrite, Ca = Calcite, H = Hematite, P = Portlandite, Q = Quartz. (B) TGA curves for native IBA and carbonated IBA
Abbreviations: AC: Ammonium carbonate; IBA: Incineration bottom ash; TGA: Thermogravimetric analysis; XRD: X-ray diffraction.
A B
Figure 2. Effect of AC concentration and time on carbonation efficiency. (a) Carbonation efficiency of IBA versus concentration of (NH ) CO based on the
3
4 2
weight loss between 550°C and 800°C of TGA (50 mL, 1 h). (b) Carbonation kinetics based on the weight loss between 550°C and 800°C of TGA. Reaction
condition: IBA (10 g), (NH ) CO (6 wt%, 50 mL), room temperature
4 2
3
Abbreviations: AC: Ammonium carbonate; IBA: Incineration bottom ash; TGA: Thermogravimetric analysis.
Volume 1 Issue 1 (2024) 5 doi: 10.36922/eer.4609