Page 90 - GPD-2-2
P. 90
Gene & Protein in Disease Verteporfin therapy for triple-negative breast cancer
A B
C D
E
F
G H
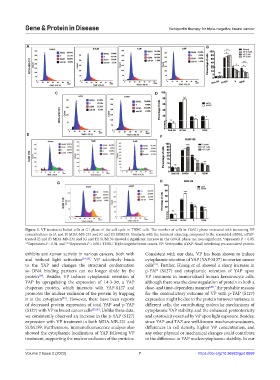
Figure 5. VP treatment halted cells at G1 phase of the cell cycle in TNBC cells. The number of cells in G0/G1 phase increased with increasing VP
concentrations in (A and B) MDA-MB-231 and (C and D) SUM159. Similarly, with the transient silencing, compared to the scrambled siRNA, siYAP-
treated (E and F) MDA-MB-231 and (G and H) SUM159 showed a significant increase in the G0/G1 phase. ns: non-significant, *represents P < 0.05,
**Represents P < 0.01, and ***Represents P < 0.001. TNBC: Triple-negative breast cancer, VP: Verteporfin; siYAP: Small interfering yes-associated protein.
exhibits anti-tumor activity in various cancers, both with Consistent with our data, VP has been shown to induce
and without light activation [19-25] . VP selectively binds cytoplasmic retention of YAP (YAP-S127) in ovarian cancer
to the YAP and changes the structural conformation cells . Further, Huang et al. showed a sharp increase in
[27]
so DNA binding partners can no longer abide by the p-YAP (S127) and cytoplasmic retention of YAP upon
protein . Besides, VP induces cytoplasmic retention of VP treatment in immortalized human keratinocyte cells,
[18]
YAP by upregulating the expression of 14-3-3σ, a YAP although there was the downregulation of protein in both a
chaperon protein, which interacts with YAP-S127 and dose- and time-dependent manner . The probable reasons
[28]
promotes the nuclear exclusion of the protein by trapping for the contradictory outcome of VP with p-YAP (S127)
it in the cytoplasm . However, there have been reports expression might be due to the protein turnover variance in
[23]
of decreased protein expression of total YAP and p-YAP different cells, the contributing molecular mechanisms of
(S127) with VP in breast cancer cells [25,26] . Unlike these data, cytoplasmic YAP stability, and the enhanced proteotoxicity
we consistently observed an increase in the p-YAP (S127) and cytotoxicity exerted by VP upon light exposure. Besides,
expression with VP treatment in both MDA-MB-231 and since YAP and TAZ are well-known mechanotransducers,
SUM159. Furthermore, immunofluorescence analysis also differences in cell density, higher VP concentration, and
showed the cytoplasmic localization of YAP following VP any other physical or mechanical changes could contribute
treatment, supporting the nuclear exclusion of the proteins. to the difference in YAP nucleocytoplasmic stability. In our
Volume 2 Issue 2 (2023) 7 https://doi.org/10.36922/gpd.0658