Page 46 - IMO-2-2
P. 46
Innovative Medicines & Omics Alzheimer’s disease and clinical trials
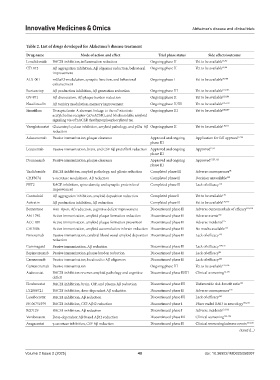
Table 2. List of drugs developed for Alzheimer’s disease treatment
Drug name Mode of action and effect Trial phase status Side effects/outcome
Lenalidomide BACE1 inhibition, inflammation reduction Ongoing phase II Yet to be available 95,96
CT1812 Aβ aggregation inhibition, Aβ oligomer reduction, behavioral Ongoing phase II Yet to be available 97,98
improvement
ALX-001 mGluR5 modulation, synaptic function, and behavioral Ongoing phase I Yet to be available 99,100
enhancement
Buntanetap Aβ production inhibition, Aβ generation reduction Ongoing phase III Yet to be available 101,102
GV-971 Aβ dissociation, Aβ plaque burden reduction Ongoing phase II Yet to be available 103,104
Nasal insulin Aβ toxicity modulation, memory improvement Ongoing phase II/III Yet to be available 105-107
Simufilam Disrupts famin A aberrant linkage to the α7 nicotinic Ongoing phase III Yet to be available 108,109
acetylcholine receptor (α7nAChR), and blocks soluble amyloid
signaling via α7nAChR that hyperphosphorylated tau
Varoglutamstat Glutaminyl cyclase inhibition, amyloid pathology, and pGlu-Aβ Ongoing phase II Yet to be available 110,111
reduction
Aducanumab Passive immunization, plaque clearance Approved and ongoing Application for full approval 17,94
phase III
Lecanemab Passive immunization, brain, and CSF Aβ protofibril reduction Approved and ongoing Approved 17,57
phase III
Donanemab Passive immunization, plaque clearance Approved and ongoing Approved 17,57,112
phase III
Thalidomide BACE1 inhibition, amyloid pathology, and gliosis reduction Completed phase III Adverse consequences 96
CHF5074 γ-secretase modulation, Aβ reduction Completed phase II Decision unavailable 113
PBT2 RAGE inhibition, spine density, and synaptic protein level Completed phase II Lack of efficacy 114
improvement
Contraloid Aβ aggregation inhibition, amyloid deposition reduction Completed phase I Yet to be available 115
Acitretin Aβ production inhibition, Aβ reduction Completed phase II Yet to be available 116,117
Bexarotene Anti-ApoE, Aβ reduction, cognitive deficit improvement Discontinued phase II Adverse outcomes/lack of efficacy 118,119
AN-1792 Active immunization, amyloid plaque formation reduction Discontinued phase II Adverse events 120
ACC-001 Active immunization, amyloid plaque formation prevention Discontinued phase II Adverse incidents 121
CAD106 Active immunization, amyloid accumulation in brain reduction Discontinued phase II No results available 122
Ponezumab Passive immunization, cerebral blood vessel amyloid deposition Discontinued phase II Lack of efficacy 123
reduction
Gammagard Passive immunization, Aβ reduction Discontinued phase II Lack of efficacy 124,125
Bapineuzumab Passive immunization, plaque burden reduction Discontinued phase II Lack of efficacy 76
Crenezumab Passive immunization, localized to Aβ oligomers Discontinued phase II Lack of efficacy 126
Gantenerumab Passive immunization Ongoing phase III Yet to be available 127,128
Atabecestat BACE1 inhibition reverses amyloid pathology and cognitive Discontinued phase II/III Clinical worsening 76,129
deficit
Elenbecestat BACE1 inhibition, brain, CSF, and plasma Aβ reduction Discontinued phase III Unfavorable risk-benefit ratio 124
LY2886721 BACE1 inhibition, dose-dependent Aβ reduction Discontinued phase II Adverse consequences 122
Lanabecestat BACE1 inhibition, Aβ reduction Discontinued phase III Lack of efficacy 123
PF-06751979 BACE1 inhibition, CSF Aβ42 reduction Discontinued phase I Pfizer ended R&D in neurology 130,131
RG7129 BACE1 inhibition, Aβ reduction Discontinued phase I Adverse incidents 121,132
Verubecestat Dose-dependent Aβ40 and Aβ42 reduction Discontinued phase III Clinical worsening 133-135
Avagacestat γ-secretase inhibition, CSF Aβ reduction Discontinued phase II Clinical worsening/adverse events 127,128
(Cont'd...)
Volume 2 Issue 2 (2025) 40 doi: 10.36922/IMO025050007