Page 30 - MI-2-2
P. 30
Microbes & Immunity Genetic therapy with HSV-1 vectors
Figure 2. Three herpes simplex virus Type I (HSV-1)-derived vectors
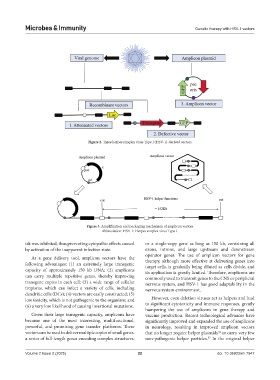
Figure 3. Amplification and packaging mechanism of amplicon vectors
Abbreviation: HSV-1: Herpes simplex virus Type I.
tsk was inhibited, thus preventing cytopathic effects caused or a single-copy gene as long as 150 kb, containing all
by activation of the inapparent infection state. exons, introns, and large upstream and downstream
operator genes. The use of amplicon vectors for gene
As a gene delivery tool, amplicon vectors have the
following advantages: (1) an extremely large transgenic therapy, although more effective at delivering genes into
target cells, is gradually being diluted as cells divide, and
capacity of approximately 150 kb DNA; (2) amplicons its application is greatly limited. Therefore, amplicons are
can carry multiple repetitive genes, thereby improving commonly used to transmit genes to the CNS or peripheral
transgene copies in each cell; (3) a wide range of cellular nervous system, and HSV-1 has good adaptability in the
tropisms, which can infect a variety of cells, including nervous system environment.
dendritic cells (DCs); (4) vectors are easily constructed; (5)
low toxicity, which is not pathogenic to the organism; and However, even deletion viruses act as helpers and lead
(6) a very low likelihood of causing insertional mutations. to significant cytotoxicity and immune responses, greatly
hampering the use of amplicons in gene therapy and
Given their large transgenic capacity, amplicons have vaccine production. Recent technological advances have
become one of the most interesting, multifunctional, significantly improved and expanded the use of amplicons
powerful, and promising gene transfer platforms. These in neurology, resulting in improved amplicon vectors
vectors can be used to deliver multiple copies of small genes, that no longer require helper plasmids or carry very few
34
a series of full-length genes encoding complex structures, non-pathogenic helper particles. In the original helper
35
Volume 2 Issue 2 (2025) 22 doi: 10.36922/mi.7947