Page 55 - TD-1-1
P. 55
Tumor Discovery Dual-targeting of cancer receptors by nanoparticles
drug release may be due to the interaction between the It had been observed that all concentrations of free
abundance of carboxyl groups in Fe O -DPN-HA-FA NPs TMX reduced both cell proliferation and viability to about
3
4
and dimethylamino groups in TMX (compound 6 shown 20% of the control after 48 h and 72 h of treatment and
in Figure 1). in the absence of any time- and concentration-dependent
inhibitory effects on MDA-MB-231 cell lines. On the
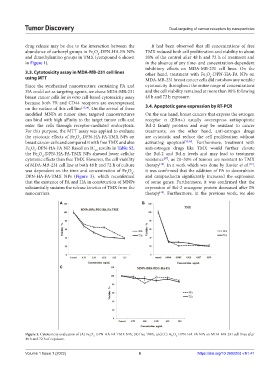
3.3. Cytotoxicity assay in MDA-MB-231 cell lines other hand, treatment with Fe O -DPN-HA-FA NPs on
3
4
using MTT MDA-MB-231 breast cancer cells did not show any notable
Since the synthesized nanostructure containing FA and cytotoxicity throughout the entire range of concentrations
HA could act as targeting agents, we chose MDA-MB-231 and the cell viability remained at more than 90% following
breast cancer cells for in vitro cell-based cytotoxicity assay 48 h and 72 h exposure.
because both FR and CD44 receptors are overexpressed
on the surface of this cell line [33,34] . On the arrival of these 3.4. Apoptotic gene expression by RT-PCR
modified MNPs at tumor sites, targeted nanostructures On the one hand, breast cancers that express the estrogen
can bind with high affinity to the target tumor cells and receptor α (ERα+) usually overexpress antiapoptotic
enter the cells through receptor-mediated endocytosis. Bcl-2 family proteins and may be resistant to cancer
For this purpose, the MTT assay was applied to evaluate treatments; on the other hand, anti-estrogen drugs
the cytotoxic effects of Fe O -DPN-HA-FA-TMX NPs on are cytostatic and reduce the cell proliferation without
3
4
breast cancer cells and compared it with free TMX and also activating apoptosis [35,36] . Furthermore, treatment with
Fe O -DPN-HA-FA NP. Based on IC results in Table S2, anti-estrogen drugs like TMX would further elevate
4
3
50
the Fe O -DPN-HA-FA-TMX NPs showed lower cellular the Bcl-2 and Bcl-x levels and may lead to treatment
3
4
cytotoxic effects than free TMX. However, the cell viability resistance , as 20–30% of tumors are resistant to TMX
[37]
of MDA-MB-231 cell line at both 48 h and 72 h of culture therapy . In a work which was done by Xavier et al. ,
[38]
[39]
was dependent on the time and concentration of Fe O - it was confirmed that the addition of FA to doxorubicin
3
4
DPN-HA-FA-TMX NPs (Figure 3), which reconfirmed and camptothecin significantly increased the expression
that the existence of FA and HA in construction of MNPs of some genes. Furthermore, it was confirmed that the
substantially sustains the release kinetics of TMX from the expression of Bcl-2 oncogene protein decreased after FA
nanocarriers. therapy . Furthermore, in the previous work, we also
[40]
A B
C
Figure 3. Cytotoxicity evaluation of (A) Fe O -DPN-HA-FA-TMX NPs, (B) free TMX, and (C) Fe O -DPN-HA-FA NPs on MDA-MB-231 cell lines after
4
3
3
4
48 h and 72 h of exposure.
Volume 1 Issue 1 (2022) 6 https://doi.org/10.36922/td.v1i1.41