Page 64 - ARNM-1-1
P. 64
Advances in Radiotherapy
& Nuclear Medicine PRaG therapy for renal pelvis carcinoma
advantages in numerous case series [9-12] . In addition, Rechallenge is a moderately infrequent therapeutic
there is currently inadequate evidence to support the use approach in the clinical management of RPC. This is
of effective treatment options for patients who undergo especially the case when conventional therapies have
disease progression after ceasing anti-PD-1 therapy. already been exhausted. In 2018, Hakozaki et al. reported
a case of an advanced non-small-cell lung cancer (NSCLC)
patient who had previously received an ICI alongside
A potent cytotoxic chemotherapy as the primary first-line
treatment. Subsequent to rechallenging the patient with
an ICI, a PR was accomplished . The report highlighted
[13]
the prospective clinical effectiveness of an ICI rechallenge
strategy in patients who undergo disease progression
after accomplishing an initial response to the same agent.
However, it is crucial to acknowledge that these instances
are moderately rare. Furthermore, the efficiency of
B rechallenge with the same PD-1 inhibitor in other tumor
types is ambiguous. The study conducted by Leighl et al.
in 2021 revealed that the amalgamation of durvalumab
and tremelimumab exhibited negligible efficacy in patients
diagnosed with advanced squamous NSCLC (sqNSCLC),
who encountered disease progression subsequent to
previous anti-PD-1 therapy . Consequently, the optimal
[14]
C strategy to administer rechallenge therapy remains
ambiguous.
Radiotherapy, traditionally regarded as a local
treatment, has recently been discovered to be a modulator
of immunotherapy. In 2018, Asna et al. reported that
radiation therapy holds the potential to enhance anti-
cancer immune responses by triggering a switch from an
immune-excluded (“cold”) pattern to an inflammatory
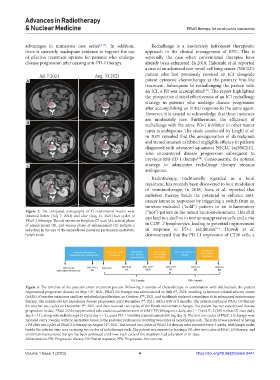
Figure 2. The computed tomography (CT) examination results were (“hot”) pattern in the tumor microenvironment. This shift
obtained before (July 7, 2021) and after (Aug 31, 2021) two cycles of can lead to a decline in immunosuppressive cells and a rise
PRaG 2.0 therapy. The red arrows on the plain CT scan (A), arterial phase +
of enhancement (B), and venous phase of enhancement (C) indicate a in CD8 T lymphocytes, leading to potential improvement
[15]
reduction in the size of the unirradiated posterior peritoneum metastatic in response to PD-1 inhibition . Dovedi et al.
lymph node. demonstrated that the PD-L1 expression of CT26 cells, a
Figure 3. The timeline of the patient’s entire treatment process. Following 3 months of chemotherapy in combination with tislelizumab, the patient
experienced progressive disease on May 13 , 2021. PRaG 2.0 therapy was administered on July 6 , 2021, resulting in immune-related adverse events
th
th
(irAEs) of reactive cutaneous capillary endothelial proliferation on October 17 , 2021, and sintilimab replaced camrelizumab in subsequent maintenance
th
therapy. The patient did not experience disease progression until December 7 , 2021, with a PFS of 5 months. The patient continued PRaG 2.0 therapy
th
for another two cycles on December 9 , 2021, and then received two cycles of sintilimab maintenance therapy. The patient has not experienced disease
th
progression to date. PRaG 2.0 therapy involved subcutaneous administration of GM-CSF (200 µg once daily, day 1 – 7) and IL-2 (200 million UI once daily,
day 8 – 14), along with radiotherapy (5 Gy/d, day 1 – 2), and a PD-1 inhibitor (camrelizumab 200 mg, day 3). The first two cycles of PRaG 2.0 therapy were
repeated every 3 weeks, with the metastatic lesion in the posterior peritoneum receiving two cycles of radiotherapy each. The patient was assessed as having
a PR after two cycles of PRaG 2.0 therapy on August 31 , 2021. The second two cycles of PRaG 2.0 therapy were repeated every 3 weeks, with lymph nodes
st
beside the inferior vena cava receiving two cycles of radiotherapy each. The patient was assessed as having a PR after two cycles of PRaG 2.0 therapy, and
sintilimab maintenance therapy has been continued until now. Each cycle of the treatment had a duration of 21 days.
Abbreviations: PD: Progressive disease; PR: Partial response; PFS: Progression-free survival.
Volume 1 Issue 1 (2023) 4 https://doi.org/10.36922/arnm.0441