Page 35 - GPD-1-1
P. 35
Gene & Protein in Disease Genetically engineered T cells in cancer immunotherapy
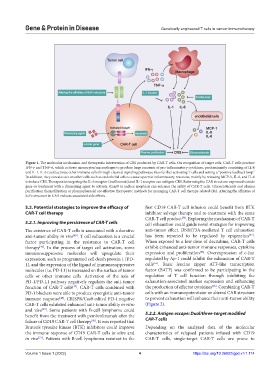
Figure 1. The molecular mechanism and therapeutic intervention of CRS produced by CAR-T cells. On recognition of target cells, CAR-T cells produce
IFN- and TNF-α, which activate monocytes/macrophages to produce large amounts of pro-inflammatory cytokines, predominantly consisting of IL-6
and IL-1. IL-6 could activate other immune cells through classical signaling pathways, thus further activating T cells and setting a “positive feedback loop.”
In addition, they can also act on other cells such as endothelial cells to cause systemic inflammatory reactions, mainly by releasing MCP-1, IL-8, and IL-6
to induce CRS. Therapeutics targeting the IL-6 receptor (tocilizumab) and IL-1 receptor can mitigate CRS. Reforming the CAR structure-expressed suicide
gene or treatment with a dimerizing agent to activate iCasp9 to induce apoptosis can enhance the safety of CAR-T cells. Glucocorticoids and plasma
purification (hemofiltration or plasmapheresis) are effective therapeutic methods for managing CAR-T cell therapy-related CRS. Altering the affinities of
ScFv structure in CAR reduces associated side effects.
5.2. Potential strategies to improve the efficacy of first CD19 CAR-T cell infusion could benefit from BTK
CAR-T cell therapy inhibitor salvage therapy and re-treatment with the same
CAR-T cell product . Exploring the mechanism of CAR-T
[52]
5.2.1. Improving the persistence of CAR-T cells cell exhaustion could guide novel strategies for improving
The existence of CAR-T cells is associated with a durative anti-tumor effect. DNMT3A-mediated T cell exhaustion
anti-tumor ability in vivo . T cell exhaustion is a crucial has been reported to be regulated by epigenetics .
[53]
[45]
factor participating in the resistance to CAR-T cell When exposed to a low dose of decitabine, CAR-T cells
[46]
therapy . In the process of target cell activation, some exhibit enhanced anti-tumor immune responses, cytokine
[54]
immunosuppressive molecules will upregulate their expression and proliferation . Overexpression of c-Jun
expression, such as programmed cell death protein 1 (PD- regulated by Ap-1 could inhibit the exhaustion of CAR-T
1), and the expression of the ligand of immunosuppressive cells . Basic leucine zipper ATF-like transcription
[55]
molecules (i.e. PD-L1) is increased on the surface of tumor factor (BATF) was confirmed to be participating in the
cells or other immune cells. Activation of the axis of regulation of T cell function through inhibiting the
PD-1/PD-L1 pathway negatively regulates the anti-tumor exhaustion-associated marker expression and enhancing
function of CAR-T cells . CAR-T cells combined with the production of effector cytokines . Combining CAR-T
[56]
[47]
PD-1 blockers were able to produce synergistic anti-tumor cells with an immunopotentiator or altered CAR structure
immune response . CRISPR/Cas9-edited PD-1 negative to prevent exhaustion will enhance their anti-tumor ability
[48]
CAR-T cells exhibited enhanced anti-tumor ability in vitro (Figure 2).
and vivo . Some patients with B-cell lymphoma could
[49]
benefit from the treatment with pembrolizumab after the 5.2.2. Antigen escape: Dual/three-target modified
failure of CD19 CAR-T cell therapy . It was reported that CAR-T cells
[50]
Bruton’s tyrosine kinase (BTK) inhibitors could improve Depending on the analyzed data of the molecular
the immune response of CD19 CAR-T cells in vitro and characteristics of relapsed patients infused with CD19
in vivo . Patients with B-cell lymphoma resistant to the CAR-T cells, single-target CAR-T cells are prone to
[51]
Volume 1 Issue 1 (2022) 4 https://doi.org/10.36922/gpd.v1i1.114