Page 36 - GPD-1-1
P. 36
Gene & Protein in Disease Genetically engineered T cells in cancer immunotherapy
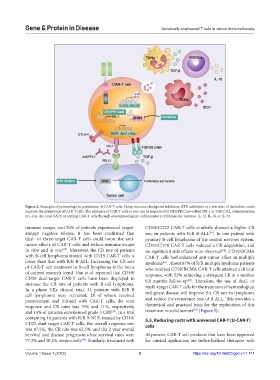
Figure 2. Strategies of promoting the persistence of CAR-T cells. Using immune checkpoint inhibitors, BTK inhibitors or a low dose of decitabine could
improve the persistence of CAR-T cells. The existence of CAR-T cells in vivo can be improved by CRISPR/Cas9-edited PD-1 or TGF-β R2, overexpression
of c-Jun, Ap-1and BATF, or arming CAR-T cells through overexpressing pro-inflammatory cytokines, for instance, IL-12, IL-18, or IL-23.
immune escape, and 50% of patients experienced target- CD19/CD22 CAR-T cells similarly showed a higher CR
antigen negative relapse. It has been confirmed that rate in patients with R/R B-ALL . In one patient with
[59]
dual- or three-target CAR-T cells could boost the anti- primary B-cell lymphoma of the central nervous system,
tumor effects of CAR-T cells and reduce immune escape CD19/CD70 CAR-T cells induced a CR acquisition, and
in vitro and in vivo . Moreover, the CR rate of patients no significant side effects were observed . CD19/BCMA
[57]
[60]
with B-cell lymphoma treated with CD19 CAR-T cells is CAR-T cells had enhanced anti-tumor effect in multiple
lower than that with R/R B-ALL. Increasing the CR rate myeloma . About 87% of R/R multiple myeloma patients
[61]
of CAR-T cell treatment in B-cell lymphoma is the focus who received CD38/BCMA CAR-T cells attained a clinical
of current research trend. Han et al. reported that CD19/ response, with 52% achieving a stringent CR at a median
CD20 dual-target CAR-T cells have been displayed to 9.0 months follow-up . Therefore, the use of dual- or
[62]
increase the CR rate of patients with B-cell lymphoma. multi-target CAR-T cells for the treatment of hematological
In a phase I/IIa clinical trial, 33 patients with R/R B
cell lymphoma were recruited, 28 of whom received malignant disease will improve the CR rate in lymphoma
pretreatment and infused with CAR-T cells, the total and reduce the recurrence rate of B-ALL. This provides a
response and CR rates was 79% and 71%, respectively, theoretical and practical basis for the exploration of this
[63]
and 14% of patients experienced grade 3 CRS . In a trial treatment in solid tumors (Figure 3).
[57]
containing 16 patients with R/R B-NHL treated by CD19/ 5.3. Reducing costs with universal CAR-T (U-CAR-T)
CD22 dual-target CAR-T cells, the overall response rate cells
was 87.5%, the CR rate was 62.5%, and the 2-year overall
survival and disease progression-free survival rates were At present, CAR-T cell products that have been approved
77.3% and 40.2%, respectively . Similarly, treatment with for clinical application are individualized therapies with
[58]
Volume 1 Issue 1 (2022) 5 https://doi.org/10.36922/gpd.v1i1.114