Page 39 - GPD-1-1
P. 39
Gene & Protein in Disease Genetically engineered T cells in cancer immunotherapy
CAR-T cells) could increase the infiltration and existence of Tregs and T cell exhaustion . Thirteen patients with
[93]
of CAR-T cells . GPC3 and mesothelin CAR-T cells metastatic castration-resistant prostate cancer received
[82]
expressing IL-7 and CCL19 produce efficient anti-tumor prostate specific antigen-directed CAR-T cells armored
response in patients with hepatoma and pancreatic cancer, with a dominant-negative TGF-βR; among the patients,
respectively . In these studies, chemokine receptor- or five patients experienced grade ≥r CRS, and 4 patients
[83]
cytokine-modified CAR-T cells showed significant tumor experienced a reduction in prostate-specific antigen
[94]
localization and anti-tumor activity in vivo (Figure 4). level . The density of TAMs is negatively correlated with
the prognosis in various types of solid tumors. Regulating
6.3. Immunosuppressive microenvironment the polarization of macrophages would reshape the tumor
In the tumor microenvironment, immunosuppressor microenvironment. For example, folate receptor β (FR) β
cells such as myeloid-derived suppressor cells, regulatory is highly expressed on the surface of macrophages, and
T cells (Tregs), tumor-associated macrophages (TAMs), FR β +TAM is related to poor outcome in pancreatic
and various cytokines contribute to tumor progression cancer. FRβ-CAR-T cells could delay tumor progression
dependent on promoting tumor invasion and metastasis through eliminating TAM, increasing pro-inflammatory
and inhibiting the T cell immune response . Overcoming monocytes and promoting endogenous tumor-specific
[84]
+
[95]
the immunosuppressive tumor microenvironment would CD8 T cell infiltration .
elevate the anti-tumor capacity of adoptively transferred 7. TCR-T cell therapy
T cells in solid tumors [85-87] . Arming CAR-T cells through
expressing pro-inflammatory cytokines such as IL-12, Tumor-specific lysis of T cells is mediated by specific
[96]
IL-18, or IL-23 can remodel the tumor microenvironment TCR-recognizing peptides and HLA complex . TCR-
and improve anti-tumor capability [88-90] . Transforming engineered T cells refers to the genetic editing of T cells
growth factor beta (TGF-β) is a vital factor for tumor in vitro by transducing TCR gene sequences (α and β
initiation and progression, which negatively regulates anti- chains) to T cells, resulting in enhanced ability of T cells
tumor immunity of T cells. It has been reported that TGF-β to specifically recognize tumor antigens on the tumor cell
could promote T cell exhaustion through up-regulating surface and effectively induce specific lysis of target tumor
PD-1 expression in cytotoxic CD8 T cells [91,92] . CRISPR/ cells. The TCR expressed by the exogenous gene binds CD3
+
Cas9-edited TGF-β R2 enhances the anti-tumor molecules in a non-covalent bond, forming a TCR-CD3
capability of CAR-T cells by reducing the transformation complex expressed on the cell membrane, which further
recognizes the HLA-antigen peptide complex. Unlike
CAR-T cells, TCR-T cells can recognize HLA peptides
derived from intracellular proteins and kill target cells in
HLA-restricted dependent manner [96-98] . The expression
of cancer/testis antigens is silent in normal tissues, except
in reproductive system, but is significantly increased in
tumor tissues, including melanoma-associated antigen
(MAGEA)-3, MAGEA-4, MART-1, gp100, and New York
esophageal squamous cell carcinoma (NY-ESO-1), which
are considered attractive targets for TCR-T cell therapy .
[99]
Approximately 37% of published clinical trials are related
to TCR-T cell therapy targeting NY-ESO-1 [100] . In 2011,
the first clinical trial of autologous NY-ESO-1-specific
TCR-T cells on advanced malignant melanoma and
synovial cell sarcoma was conducted; 4 of 6 synovial cell
sarcoma patients and 5 of 11 melanoma patients achieved
ORR, and with a CR after one year was observed in two
melanoma patients . MAGE-A4 TCR-T cells produced
[5]
significant anti-tumor ability in patients with esophageal
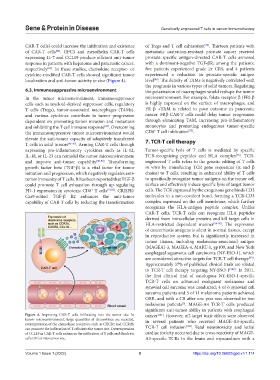
Figure 4. Improving CAR-T cells infiltrating into the tumor site. In cancer [101] . However, off-target toxic effects were observed
tumor microenvironment, large quantities of chemokines are secreted, in several patients who received MAGE-A3-specific
overexpression of the chemokine receptors, such as CXCR2 and CCR2b, [102]
can promote the infiltration of T cells into the tumor site. Overexpression TCR-T cell infusion . Fatal neurotoxicity and lethal
of CCL19 in CAR-T cells enhances the infiltration of T cells and dendritic cardiac toxicity occurred due to cross-reactivity of MAGE-
cells (DCs) into tumor site. A3-specific TCRs in the brain and myocardium with a
Volume 1 Issue 1 (2022) 8 https://doi.org/10.36922/gpd.v1i1.114