Page 38 - GPD-1-1
P. 38
Gene & Protein in Disease Genetically engineered T cells in cancer immunotherapy
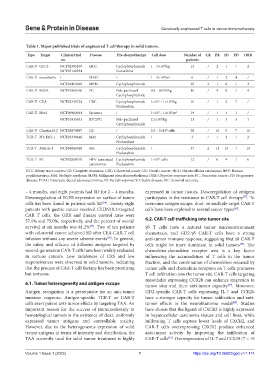
Table 1. Major published trials of engineered T cell therapy in solid tumors.
Type Target Clinical trial Disease Pre‑chemotherapy Cell dose Number of CR PR SD PD ORR
no. patients
CAR-T GPC3 NCT02395250 HCC Cyclophosphamide 1 – 3×10 /kg 13 / 2 1 / 2
5
NCT03146234 fludarabine
CAR-T mesothelin / PDAC / 1 – 3×10 /m 2 6 / / 2 4 /
8
NCT02414269 MPM Cyclophosphamide 25 2 / 8 / 2
CAR-T EGFR NCT01869166 PC Nab-paclitaxel 0.3 – 60 M/kg 16 / 4 8 / 4
Cyclophosphamide
5
8
CAR-T CEA NCT02349724 CRC Cyclophosphamide 1×10 – 1×10 /kg 10 / / 2 7 /
Fludarabine
CAR-T Her2 NCT00902044 Sarcoma / 1×10 – 1×10 /m 2 19 / / 4 1 /
4
8
NCT01935843 BTC/PC Nab-paclitaxel 2.1×10 /kg 11 / 1 5 5 1
6
Cyclophosphamide
CAR-T Claudin18.2 NCT03874897 GC / 2.5 – 5×10 cells 28 / 16 5 7 16
8
TCR-T NY-ESO-1 NCT03399448 MM Cyclophosphamide / 3 / / 2 / 2
Fludarabine
TCR-T MAGE-4 NCT04044768 SSC Cyclophosphamide / 37 2 11 15 / 13
Fludarabine
TCR-T E7 NCT02858310 HPV-associated Cyclophosphamide 1×10 cells 12 / 6 4 / 6
11
carcinomas Fludarabine
BTC: Biliary tract cancers; CR: Complete remission; CRC: Colorectal cancer; GC: Gastric cancer; HCC: Hepatocellular carcinoma; HPV: Human
papillomavirus; MM: Multiple myeloma; MPM: Malignant pleural mesothelioma; ORR: Objective response rate; PC: Pancreatic cancer; PD: Progressive
disease; PDAC: Pancreatic ductal adenocarcinoma; PR: Partial response; SD: Stable disease; SSC: Synovial sarcoma.
– 4 months, and eight patients had SD for 2 – 4 months. expressed in tumor tissues. Downregulation of antigens
Downregulation of EGFR expression on surface of tumor participates in the resistance to CAR-T cell therapy . To
[77]
cells has been found in patients with SD . Twenty-eight overcome antigen escape, dual- or multiple-target CAR-T
[74]
patients with gastric cancer received CLDN18.2-targeted cells have been explored in several cancer types .
[63]
CAR T cells, the ORR and disease control rates were
57.1% and 75.0%, respectively, and the percent of overall 6.2. CAR-T cell trafficking into tumor site
survival at six months was 81.2% . Two of ten patients γδ T cells have a natural tumor microenvironment
[75]
with colorectal cancer achieved SD after CEA-CAR-T cell chemotaxis, and GD2-γδ CAR-T cells have a strong
[76]
infusion without any severe adverse events . In general, anti-tumor immune response, suggesting that γδ CAR-T
the safety and efficacy of different antigens targeted by cells might be more dominant in solid tumors . The
[78]
second-generation CAR-T cells have been widely evaluated chemokine-chemokine receptor axis is a key factor
in various cancers. Low incidences of CRS and low influencing the accumulation of T cells in the tumor
responsiveness were observed in solid tumors, indicating fraction, and the combination of chemokines released by
that the process of CAR-T cell therapy has been promising tumor cells and chemokine receptors on T cells promotes
but tortuous. T cell infiltration into the tumor site. CAR-T cells targeting
mesothelin expressing CCR2b can enhance migration to
6.1. Tumor heterogeneity and antigen escape tumor sites and their anti-tumor capacity . Moreover,
[79]
Antigen recognition is a prerequisite for an anti-tumor GD2-specific CAR-T cells expressing IL-7 and CCR2b
immune response. Antigen-specific TCR-T or CAR-T have a stronger capacity for tumor infiltration and anti-
cells exert potent anti-tumor effects by targeting TAA. An tumor effects in the neuroblastoma model . Studies
[80]
important reason for the success of immunotherapy in have shown that the ligand of CXCR2 is highly expressed
hematological tumors is the existence of clear, uniformly in hepatocellular carcinoma tissues and cell lines, while
expressed tumor antigens and controllable toxicity. infiltrating T cells express lower levels of CXCR2, and
However, due to the heterogeneous expression of solid CAR-T cells overexpressing CXCR2 produce enhanced
tumor antigens in terms of intensity and distribution, the anti-tumor activity by improving the infiltration of
TAA currently used for solid tumor treatment is highly CAR-T cells . Overexpression of IL-7 and CCL19 (7 × 19
[81]
Volume 1 Issue 1 (2022) 7 https://doi.org/10.36922/gpd.v1i1.114