Page 28 - GPD-3-4
P. 28
Gene & Protein in Disease GLUT5 in cancer development and therapy
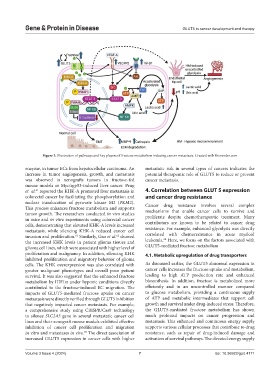
Figure 3. Illustration of pathways and key players of fructose metabolism inducing cancer metastasis. Created with Biorender.com
enzyme, in tumor ECs from hepatocellular carcinoma. An metastatic risk in several types of cancers indicates the
increase in tumor angiogenesis, growth, and metastasis potential therapeutic role of GLUT5 to reduce or prevent
was observed in xenografts tumors in fructose-fed cancer metastasis.
mouse models or Myc/sgp53-induced liver cancer. Peng
61
et al. reported the KHK-A promoted liver metastasis in 4. Correlation between GLUT 5 expression
colorectal cancer by facilitating the phosphorylation and and cancer drug resistance
nuclear translocation of pyruvate kinase M2 (PKM2). Cancer drug resistance involves several complex
This process enhances fructose metabolism and supports mechanisms that enable cancer cells to survive and
tumor growth. The researchers conducted in vivo studies proliferate despite chemotherapeutic treatment. Many
in mice and in vitro experiments using colorectal cancer
cells, demonstrating that elevated KHK-A levels increased contributors are known to be related to cancer drug
metastasis, while silencing KHK-A reduced cancer cell resistance. For example, enhanced glycolysis was directly
invasion and proliferation. Similarly, Gao et al. showed correlated with chemoresistance in acute myeloid
62
61
64
the increased KHK levels in patient glioma tissues and leukemia. Here, we focus on the factors associated with
glioma cell lines, which were associated with higher level of GLUT5-mediated fructose metabolism
proliferation and malignancy. In addition, silencing KHK 4.1. Metabolic upregulation of drug transporters
inhibited proliferation and migratory behavior of glioma
cells. The KHK overexpression was also correlated with As discussed earlier, the GLUT5 abnormal expression in
greater malignant phenotypes and overall poor patient cancer cells increases the fructose uptake and metabolism,
survival. It was also suggested that the enhanced fructose leading to high ATP production rate and enhanced
metabolism by HIF1α under hypoxic conditions directly biosynthesis. In addition, fructose is metabolized more
contributed to the fructose-induced EC migration. The efficiently and in an uncontrolled manner compared
impacts of GLUT5-mediated fructose uptake on cancer to glucose metabolism, providing a continuous supply
metastasis were directly verified through GLUT5 inhibition of ATP and metabolic intermediates that support cell
that negatively impacted cancer metastasis. For example, growth and survival under drug-induced stress. Therefore,
a comprehensive study using CRISPR/Cas9 technology the GLUT5-mediated fructose metabolism has shown
to silence SLC2A5 gene in several metastatic cancer cell much profound impacts on cancer progression and
lines and their xenograft mouse models exhibited effective metastasis. This enhanced and continuous energy supply
inhibition of cancer cell proliferation and migration supports various cellular processes that contribute to drug
in vitro and metastases in vivo. The direct association of resistance, such as repair of drug-induced damage and
63
increased GLUT5 expression in cancer cells with higher activation of survival pathways. The elevated energy supply
Volume 3 Issue 4 (2024) 7 doi: 10.36922/gpd.4171