Page 105 - MI-1-1
P. 105
Microbes & Immunity Factors associated with response to T-VEC
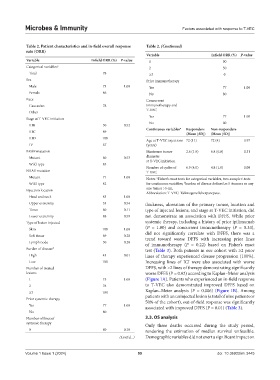
Table 2. Patient characteristics and in‑field overall response Table 2. (Continued)
rate (ORR)
Variable Infield ORR (%) P‑value
Variable Infield ORR (%) P‑value 1 90
Categorical variables a 2 50
Total 78 ≥3 0
Sex Prior immunotherapy
Male 73 1.00 Yes 77 1.00
Female 86 No 80
Race Concurrent
Caucasian 78 immunotherapy and
T-VEC
Other -
Yes 77 1.00
Stage at T-VEC initiation
No 80
IIIB 50 0.52
IIIC 89 Continuous variables a Responders Non‑responders
(Mean [SD]) (Mean [SD])
IIID 100
Age at T-VEC injections 72 (11) 72 (4) 0.97
IV 67 (years)
BRAF mutation Maximum tumor 2.6 (1.9) 6.8 (6.8) 0.31
Mutant 60 0.53 diameter
at T-VEC initiation
Wild-type 85
Number of cycles of 6.9 (4.0) 4.8 (1.0) 0.09
NRAS mutation T-VEC
Mutant 71 1.00 Notes: Fisher’s exact tests for categorical variables, two-sample t-tests
a
Wild-type 82 for continuous variables; burden of disease defined as 5+tumors or any
b
Injection location one tumor >5 cm.
Abbreviation: T-VEC: Talimogene laherparepvec.
Head and neck 83 1.00
Upper extremity 33 0.54 thickness, ulceration of the primary tumor, location and
Torso 60 0.11 type of injected lesions, and stage at T-VEC initiation, did
Lower extremity 88 0.59 not demonstrate an association with DFFS. While prior
Type of lesion injected systemic therapy, including a history of prior ipilimumab
Skin 100 1.00 (P = 1.00) and concurrent immunotherapy (P = 0.33),
Soft tissue 69 0.28 did not significantly correlate with DFFS, there was a
trend toward worse DFFS with increasing prior lines
Lymph node 50 0.20 of immunotherapy (P = 0.22) based on Fisher’s exact
Burden of disease b test (Table 3). Both patients in our cohort with ≥2 prior
High 43 0.01 lines of therapy experienced disease progression (100%).
Low 100 Increasing lines of ICI were also associated with worse
Number of treated DFFS, with >2 lines of therapy demonstrating significantly
lesions worse DFFS (P = 0.02) according to Kaplan–Meier analysis
1 75 1.00 (Figure 1A). Patients who experienced an in-field response
2 78 to T-VEC also demonstrated improved DFFS based on
≥3 100 Kaplan–Meier analysis (P = 0.006) (Figure 1B). Among
Prior systemic therapy patients with an uninjected lesion (a total of nine patients or
50% of the cohort), out-of-field response was significantly
Yes 77 1.00 associated with improved DFFS (P = 0.01) (Table 3).
No 80
Number of lines of 3.3. OS analysis
systemic therapy Only three deaths occurred during the study period,
0 80 0.18 rendering the estimation of median survival unfeasible.
(Cont'd...) Demographic variables did not exert a significant impact on
Volume 1 Issue 1 (2024) 99 doi: 10.36922/mi.3445