Page 108 - MI-1-1
P. 108
Microbes & Immunity Factors associated with response to T-VEC
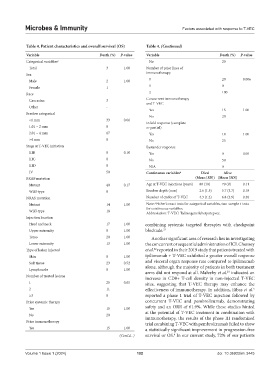
Table 4. Patient characteristics and overall survival (OS) Table 4. (Continued)
Variable Death (%) P‑value Variable Death (%) P‑value
Categorical variables a No 20
Total 3 1.00 Number of prior lines of
Sex immunotherapy
Male 2 1.00 0 20 0.006
Female 1 1 0
Race 2 100
Caucasian 3 Concurrent immunotherapy
and T-VEC
Other -
Yes 15 1.00
Breslow categorical
No 20
<1 mm 33 0.06 Infield response (complete
1.01 – 2 mm 0 or partial)
2.01 – 4 mm 67 Yes 14 1.00
>4 mm 0 No 25
Stage at T-VEC initiation Bystander response
IIIB 0 0.10 Yes 0 0.05
IIIC 0 No 50
IIID 0 N/A 0
IV 50 Continuous variables a Died Alive
BRAF mutation (Mean [SD]) (Mean [SD])
Mutant 40 0.17 Age at T-VEC injections (years) 80 (10) 70 (9) 0.11
Wild-type 8 Breslow depth (mm) 2.6 (1.5) 5.7 (3.7) 0.18
NRAS mutation Number of cycles of T-VEC 4.3 (1.2) 6.8 (3.9) 0.30
a
Mutant 14 1.00 Note: Fisher’s exact tests for categorical variables, two-sample t-tests
for continuous variables.
Wild-type 18
Abbreviation: T-VEC: Talimogene laherparepvec.
Injection location
Head and neck 17 1.00 combining systemic targeted therapies with checkpoint
Upper extremity 0 1.00 blockade. 21
Torso 20 1.00 Another significant area of research lies in investigating
Lower extremity 13 1.00 the concurrent or sequential administration of ICI. Chesney
22
Type of lesion injected et al. reported in their 2019 study that patients treated with
Skin 0 1.00 ipilimumab + T-VEC exhibited a greater overall response
and visceral organ response rate compared to ipilimumab
Soft tissue 23 0.52
Lymph node 0 1.00 alone, although the majority of patients in both treatment
23
Number of treated lesions arms did not respond at all. Malvehy et al. indicated an
increase in CD8+ T-cell density in non-injected T-VEC
1 25 0.65 sites, suggesting that T-VEC therapy may enhance the
2 11 effectiveness of immunotherapy. In addition, Ribas et al.
5
≥3 0 reported a phase 1 trial of T-VEC injection followed by
Prior systemic therapy concurrent T-VEC and pembrolizumab, demonstrating
Yes 15 1.00 safety and an ORR of 61.9%. While these studies hinted
at the potential of T-VEC treatment in combination with
No 20
Prior immunotherapy immunotherapy, the results of the phase III randomized
trial combining T-VEC with pembrolizumab failed to show
Yes 15 1.00 a statistically significant improvement in progression-free
4
(Cont'd...) survival or OS. In our current study, 72% of our patients
Volume 1 Issue 1 (2024) 102 doi: 10.36922/mi.3445