Page 107 - MI-1-1
P. 107
Microbes & Immunity Factors associated with response to T-VEC
A B
C
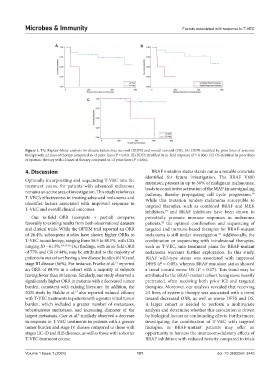
Figure 1. The Kaplan–Meier analysis for disease failure-free survival (DDFS) and overall survival (OS). (A) DDFS stratified by prior lines of systemic
therapy with ≥2 lines of therapy compared to <2 prior lines (P = 0.02). (B) DDFS stratified by in-field response (P = 0.006). (C) OS stratified by prior lines
of systemic therapy with ≥2 lines of therapy compared to <2 prior lines (P = 0.06).
4. Discussion BRAF mutation status stands out as a notable covariate
identified for future investigation. The BRAF V600
Optimally incorporating and sequencing T-VEC into the mutation, present in up to 50% of malignant melanomas,
treatment course for patients with advanced melanoma leads to constitutive activation of the MAP kinase signaling
remains an active area of investigation. This study reinforces pathway, thereby propagating cell cycle progression.
18
T-VEC’s effectiveness in treating advanced melanoma and While this mutation renders melanoma susceptible to
identifies factors associated with improved response to targeted therapies, such as combined BRAF and MEK
T-VEC and overall clinical outcomes. inhibitors, and BRAF inhibitors have been shown to
19
Our in-field ORR (complete + partial) compares potentially promote immune responses in melanoma
favorably to existing results from both observational datasets patients, the optimal combination and sequencing of
20
and clinical trials. While the OPTiM trial reported an ORR targeted and immune-based therapies for BRAF-mutant
of 26.4%, subsequent studies have shown higher ORRs to melanoma is still under investigation. Additionally, the
21
T-VEC monotherapy, ranging from 56.5 to 88.5%, with CRs combination or sequencing with intralesional therapies,
ranging 20 – 61.5%. 6,9,13-16 Our findings, with an in-field ORR such as T-VEC, into treatment plans for BRAF-mutant
of 77% and CR of 44%, may be attributed to the majority of melanoma warrants further exploration. In this study,
patients in our cohort having a low disease burden (61%) and BRAF wild-type status was associated with improved
stage III disease (66%). For instance, Franke et al. reported DFFS (P = 0.05), whereas BRAF mutation status showed
13
an ORR of 88.5% in a cohort with a majority of subjects a trend toward worse OS (P = 0.17). This trend may be
having fewer than 10 lesions. Similarly, our study observed a attributed to the BRAF-mutant cohort being more heavily
significantly higher ORR in patients with a decreased tumor pretreated, often receiving both prior ICI and targeted
burden, consistent with existing literature. In addition, the therapies. Moreover, our analysis revealed that receiving
2021 study by Stahlie et al. also reported reduced efficacy ≥2 lines of systemic therapy was associated with a trend
17
with T-VEC treatments in patients with a greater initial tumor toward decreased ORR, as well as worse DFFS and OS.
burden, which included a greater number of metastases, A larger cohort is needed to perform a multivariate
subcutaneous metastases, and increasing diameter of the analysis and determine whether this association is driven
largest metastasis. Carr et al. similarly observed a decrease by biological factors or confounding effects. Furthermore,
6
in response to T-VEC treatments in patients with a greater investigating the combination of T-VEC with targeted
tumor burden and stage IV disease compared to those with therapies in BRAF-mutant patients may offer an
stages IIC–D and IIIB diseases, as well as those with a shorter opportunity to harness the immunomodulatory effects of
T-VEC treatment course. BRAF inhibition with reduced toxicity compared to trials
Volume 1 Issue 1 (2024) 101 doi: 10.36922/mi.3445