Page 56 - MSAM-1-1
P. 56
Materials Science in Additive Manufacturing From 3D printed molds to bioprinted scaffolds
material. This proved to pose a problem when removing a Acellular human ear constructs were 3D bioprinted
print from the mold post-printing. It also disrupted material with peptide-based bioink to form a soft bioink scaffold
flow in the bottom layers as the structure details would be (Figure 4A). Automated time-dependent extrusion of
more defined. To combat this, Formlabs® elastic 50A resin the bioink enabled consistent flow throughout printing,
was used. This facilitated the removal process considerably. which was essential for the construct to take a well-formed
It is also worth mentioning that this resin is biocompatible, shape. G-code optimization also facilitated a collision-free
which further supports cellular bioprinting. However, with printing experience. Post-printing, the constructs were
the flexible mold, it was observed that the bottom printed evaluated for shape fidelity and cell viability. In terms
layer had a tendency to attach to the base of the mold, which of mechanical structure, the 3D bioprinted full human
was undesirable, as it would negatively affect print quality. ear scaffold was found to maintain its shape and easily
This was resolved by reducing the mold thickness to create reconstruct the complex geometry of the model despite its
a pop-in, pop-out effect. This further improved the removal small-scale size.
process and allowed the structure to maintain its shape. The 3D bioprinting of cellular scaffolds was done in the
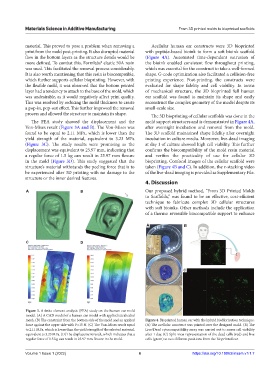
The FEA study showed the displacement and the mold support structures and is demonstrated in Figure 4A,
Von-Mises result (Figure 3A and B). The Von-Mises was after overnight incubation and removal from the mold.
found to be equal to 2.11 MPa, which is lower than the The 3D scaffold maintained shape fidelity after overnight
yield strength of the material, equivalent to 3.23 MPa incubation in culture media. Moreover, live-dead imaging
(Figure 3C). The study results were promising as the at day 1 of culture showed high cell viability. This further
displacement was equivalent to 25.97 mm, indicating that confirms the biocompatibility of the mold resin material
a regular force of 1.5 kg can result in 25.97 mm flexure and verifies the practicality of use for cellular 3D
in the mold (Figure 3D). This study suggested that the bioprinting. Confocal images of the cellular scaffold were
structure’s material withstands the peeling force that is to taken (Figure 4B and C). In addition, the z-stacking video
be experienced after 3D printing with no damage to the of the live-dead imaging is provided as Supplementary File.
structure or the inner desired features.
4. Discussion
A B Our proposed hybrid method, “From 3D Printed Molds
to Scaffolds,” was found to be an effective, cost-efficient
technique to fabricate complex 3D cellular structures
with soft bioinks. Other methods include the application
of a thermo reversible biocompatible support to enhance
A B
C D
C
Figure 3. A finite element analysis (FEA) study on the human ear mold
model. (A) A CAD model of a human ear model with applied tetrahedral
mesh. (B) The constraint from the bottom side of the mold and an applied Figure 4. Bioprinted human ear with the hybrid biofabrication technique.
force against the upper side with F=15 N. (C) The Von-Mises result equal (A) The acellular construct was printed over the designed mold. (B) The
to 2.11 MPa, which is lower than the yield strength of the selected material, Live/Dead cytocompatibility assay was carried out to assess cell viability
equivalent to 3.23 MPa. (D) The displacement result, which indicates that a after 1 day. (C) Split-view representation of the dead cells (red) and live
regular force of 1.5 kg can result in 25.97 mm flexure in the mold. cells (green) at two different positions from the bioprinted ear.
Volume 1 Issue 1 (2022) 6 https://doi.org/10.18063/msam.v1i1.7