Page 14 - MSAM-1-3
P. 14
Materials Science in Additive Manufacturing Biodegradable sustainable electronics
(mono-Si) , amorphous silicon (a-Si) , polycrystalline germanium alloys have also shown dissolution in varying
[69]
[68]
silicon (poly-Si) , and silicon alloys . The various Si pH and temperature ranges [73,75] . Increased pH led to
[70]
[71]
forms used in the present day do not degrade due to the reactions that produced metagermanic acid (H GeO ) on
2
3
formation of a native oxide layer on top, which renders hydrolysis.
them chemically inert. Research has been focused toward Inorganic oxides such as magnesium oxide (MgO),
increasing the hydrolysis rate of Si by reducing their zinc oxide (ZnO), and silicon dioxide (SiO ) have also
2
thickness and aspect ratio to make nanostructures . been explored in biodegradable electronics due to their
[72]
The hydrolysis of Si in water results in the formation superior thermal and chemical stability. The rationale is
[72]
of orthosilicic acid (Si(OH) ) . Most of the work to keep the material layers thin for better dissolution, as
4
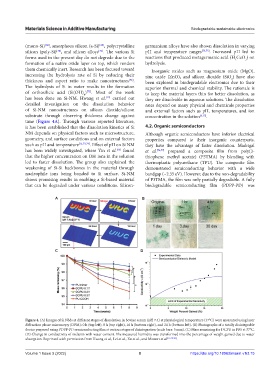
has been done on Si-NM. Hwang et al. carried out they are dissolvable in aqueous solutions. The dissolution
[12]
detailed investigation on the dissolution behavior rates depend on many physical and chemicals properties
of Si-NM nanostructures on silicon dioxide/silicon and external factors such as pH, temperatures, and ion
substrate through observing thickness change against concentration in the solution [9,17] .
time (Figure 4A). Through various reported literature,
it has been established that the dissolution kinetics of Si 4.2. Organic semiconductors
NM depends on physical factors such as microstructure, Although organic semiconductors have inferior electrical
geometry, and surface conditions and on external factors properties compared to their inorganic counterparts,
such as pH and temperature [12,73,74] . Effect of pH on Si NM they have the advantage of faster dissolution. Madrigal
has been widely investigated, where Yin et al. found et al. [76,77] prepared a composite film from poly(3-
[55]
that the higher concentration on OH ions in the solution thiophene methyl acetate) (P3TMA) by blending with
-
led to faster dissolution. The group also explained the thermoplastic polyurethane (TPU). The composite film
weakening of Si-Si backbones in the material through demonstrated semiconducting behavior with a wide
nucleophile ions being bonded to Si surface. Si-NM bandgap (~2.35 eV). However, due to the non-degradability
shows promising results in enabling a Si-based material of P3TMA, the film was only partially degradable. A fully
that can be degraded under various conditions. Silicon- biodegradable semiconducting film (PDPP-PD) was
A B
C D
Figure 4. (A) Images of Si NMs at different stages of dissolution in bovine serum (pH 7.4) at physiological temperature (37°C) were measured using laser
diffraction phase microscopy (DPM): 0 h (top left), 8 h (top right), 16 h (bottom right), and 24 h (bottom left). (B) Photographs of a totally disintegrable
device prepared using PDPP-PD semiconducting film at various stages of disintegration (scale bars: 5 mm). (C) Mass remaining for DCPU in PBS at 37°C.
(D) Change in conductivity of melanin with water content. The measured humidity was transformed into the percentage of weight gained due to water
absorption. Reprinted with permission from Hwang et al., Lei et al., Xu et al., and Mostert et al .[12,78-80] .
Volume 1 Issue 3 (2022) 8 https://doi.org/10.18063/msam.v1i3.15