Page 103 - AIH-2-2
P. 103
Artificial Intelligence in Health Cirrhosis prediction in hepatitis C
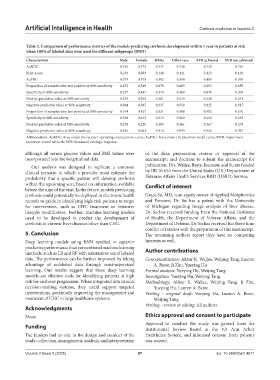
Table 3. Comparison of performance metrics of the models predicting cirrhosis development within 1 year in patients at risk
when 100% of labeled data were used for different subgroups (RNN)
Characteristic Male Female White Other race SVR achieved SVR not achieved
AuROC 0.745 0.772 0.737 0.748 0.742 0.746
Brier score 0.130 0.093 0.140 0.121 0.123 0.129
AuPRC 0.373 0.373 0.392 0.356 0.409 0.370
Proportion of samples who test positive at 90% sensitivity 0.672 0.549 0.678 0.665 0.693 0.659
Specificity at 90% sensitivity 0.377 0.487 0.376 0.380 0.876 0.393
Positive predictive value at 90% sensitivity 0.239 0.201 0.261 0.219 0.220 0.242
Negative predictive value at 90% sensitivity 0.944 0.947 0.937 0.950 0.925 0.947
Proportion of samples who test positive at 80% sensitivity 0.514 0.427 0.531 0.500 0.503 0.510
Specificity at 80% sensitivity 0.548 0.613 0.534 0.562 0.552 0.552
Positive predictive value at 80% sensitivity 0.278 0.225 0.295 0.261 0.267 0.278
Negative predictive value at 80% sensitivity 0.926 0.941 0.914 0.935 0.924 0.927
Abbreviations: AuROC: Area under the receiver operating characteristic curve; AuPRC: Area under the precision-recall curve; RNN: Supervised
recurrent neural network; SVR: Sustained virologic response.
although all serum glucose values and BMI values were of the data; preparation, review, or approval of the
incorporated into the longitudinal data. manuscript; and decision to submit the manuscript for
Our analysis was designed to replicate a common publication. Drs. Waljee, Beste, Ioannou, and Su are funded
clinical scenario in which a provider must estimate the by IIR 16-024 from the United States (U.S.) Department of
probability that a specific patient will develop cirrhosis Veterans Affairs Health Services R&D (HSRD) Service.
within the upcoming year, based on information available Conflict of interest
before the time of the visit. In the future, models predicting
cirrhosis could potentially be deployed in electronic health Grace Su, MD, is an equity owner of Applied Morphomics
records to guide in identifying high-risk patients to target and Prenovo. Dr. Su has a patent with the University
for intervention, such as CHC treatment or intensive of Michigan regarding image analysis of liver disease.
lifestyle modification. Further, machine learning models Dr. Su has received funding from the National Institutes
need to be developed to predict the development of of Health, the Department of Veteran Affairs, and the
cirrhosis in chronic liver diseases other than CHC. Department of Defense. Dr. Su has received but there is no
conflict of interest with the preparation of this manuscript.
5. Conclusion The remaining authors report they have no competing
Deep learning models using RNN resulted in superior interests as well.
predictive performance than conventional machine learning
methods, such as LR and RF with substantive use of labeled Author contributions
data. The performance can be further improved by taking Conceptualization: Akbar K. Waljee, Weijing Tang, Lauren
advantage of unlabeled data through semi-supervised A. Beste, Ji Zhu, Yueying Hu
learning. Our results suggest that these deep learning Formal analysis: Yueying Hu, Weijing Tang
models are effective tools for identifying patients at high Investigation: Yueying Hu, Weijing Tang
risk for cirrhosis progression. When integrated into clinical Methodology: Akbar K. Waljee, Weijing Tang, Ji Zhu,
decision-making systems, they could support targeted Yueying Hu, Lauren A. Beste
interventions, potentially improving the management and Writing – original draft: Yueying Hu, Lauren A. Beste,
treatment of CHC in large healthcare systems. Weijing Tang
Writing – review & editing: All authors
Acknowledgments
None. Ethics approval and consent to participate
Funding Approval to conduct the study was gained from the
Institutional Review Board at the VA Ann Arbor
The funders had no role in the design and conduct of the Healthcare System, and informed consent from patients
study; collection, management, analysis, and interpretation was waived.
Volume 2 Issue 2 (2025) 97 doi: 10.36922/aih.4671