Page 47 - GTM-2-2
P. 47
Global Translational Medicine FTO gene and obesity
70
60
50
40
30
20
10
0
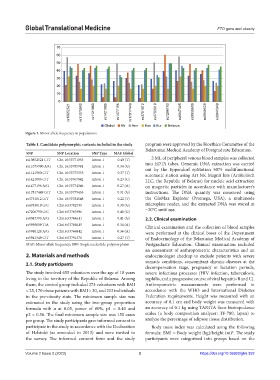
rs10852521 rs11075990 rs1121980 rs1421085 rs1477196 rs17817449 rs3751812 rs4783819 rs7206790 rs8047395 rs9939609 rs9940128 rs9941349
Global Afr Amr Eas Eur Belarus
Figure 1. Minor allele frequency in populations.
Table 1. Candidate polymorphic variants included in the study program were approved by the Bioethics Committee of the
Belarusian Medical Academy of Postgraduate Education.
SNP SNP Location SNP Type MAF Global
rs10852521 C/T Chr. 16:53771053 Intron 1 0.40 (T) 2 ML of peripheral venous blood samples was collected
rs11075990 A/G Chr. 16:53785981 Intron 1 0.34 (G) into EDTA tubes. Genomic DNA extraction was carried
out by the Eppendorf epMotion 5075 multifunctional
rs1121980 C/T Chr. 16:53775335 Intron 1 0.37 (T) automatic station using Art NK Magnit kits (ArtBioTech
rs1421085 C/T Chr. 16:53767042 Intron 1 0.23 (C) LLC, the Republic of Belarus) for nucleic acid extraction
rs1477196 A/G Chr. 16:53774346 Intron 1 0.27 (A) on magnetic particles in accordance with manufacturer’s
rs17817449 G/T Chr. 16:53779455 Intron 1 0.31 (G) instructions. The DNA quantity was measured using
rs3751812 G/T Chr. 16:53784548 Intron 1 0.22 (T) the GloMax Explorer (Promega, USA), a multimode
rs4783819 G/C Chr16:53782735 Intron 1 0.30 (G) microplate reader, and the extracted DNA was stored at
rs7206790 G/C Chr16:53763996 Intron 1 0.40 (G) −20°C until use.
rs8047395 A/G Chr16:53764611 Intron 1 0.41 (G) 2.2. Clinical examination
rs9939609 T/A Chr16:53786615 Intron 1 0.34 (A) Clinical examination and the collection of blood samples
rs9940128 A/G Chr16:53766842 Intron 1 0.36 (A) were performed at the clinical bases of the Department
rs9941349 C/T Chr16:53791576 Intron 1 0.27 (T) of Endocrinology of the Belarusian Medical Academy of
MAF: Minor allele frequency; SNP: Single nucleotide polymorphism Postgraduate Education. Clinical examination included
an assessment of anthropometric characteristics and an
2. Materials and methods endocrinologist checkup to exclude patients with severe
somatic conditions, concomitant chronic diseases at the
2.1. Study participants
decompensation stage, pregnancy or lactation periods,
The study involved 655 volunteers over the age of 18 years severe infectious processes (HIV infection, tuberculosis,
living in the territory of the Republic of Belarus. Among syphilis, and a progressive course of viral hepatitis B and C).
them, the control group included 273 volunteers with BMI Anthropometric measurements were performed in
< 25, 170 obese patients with BMI > 30, and 212 individuals accordance with the WHO and International Diabetes
in the pre-obesity state. The minimum sample size was Federation requirements. Height was measured with an
estimated in the study using the two-group proportion accuracy of 0.1 cm and body weight was measured with
formula with α at 0.05, power of 80%, p1 = 0.40 and an accuracy of 0.1 kg using TANITA floor bioimpedance
p2 = 0.56. The final minimum sample size was 150 cases scales (a body composition analyzer; TF-780, Japan) to
per group. The study participants gave informed consent to analyze the percentage of adipose tissue distribution.
participate in the study in accordance with the Declaration Body mass index was calculated using the following
of Helsinki (as amended in 2013) and were invited to formula: BMI = Body weight (kg)/height (m) . The study
2
the survey. The informed consent form and the study participants were categorized into groups based on the
Volume 2 Issue 2 (2023) 3 https://doi.org/10.36922/gtm.352