Page 28 - IMO-1-1
P. 28
Innovative Medicines & Omics Antioxidant nanomedicines for therapies
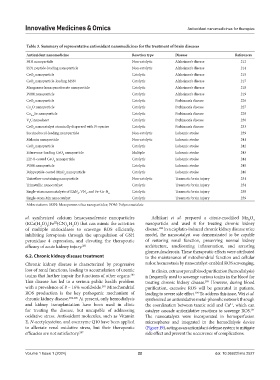
Table 3. Summary of representative antioxidant nanomedicines for the treatment of brain diseases
Antioxidant nanomedicine Reaction type Disease References
PdH nanoparticle Non-catalytic Alzheimer’s disease 212
SS31 peptide-loading nanoparticle Non-catalytic Alzheimer’s disease 214
CeO nanoparticle Catalytic Alzheimer’s disease 215
2
CeO nanoparticle-loading MSN Catalytic Alzheimer’s disease 217
2
Manganese hexacyanoferrate nanoparticle Catalytic Alzheimer’s disease 218
POM nanoparticle Catalytic Alzheimer’s disease 219
CeO nanoparticle Catalytic Parkinson’s disease 226
2
Cu O nanoparticle Catalytic Parkinson’s disease 227
x
Cu Se nanoparticle Catalytic Parkinson’s disease 229
2−x
V C nanosheet Catalytic Parkinson’s disease 230
2
CeO nanocatalyst atomically dispersed with Pt species Catalytic Parkinson’s disease 233
2
Succinobucol-loading nanoparticle Non-catalytic Ischemic stroke 239
Melanin nanoparticle Non-catalytic Ischemic stroke 241
CeO nanoparticle Catalytic Ischemic stroke 242
2
Edaravone-loading CeO nanoparticle Multiple Ischemic stroke 243
2
ZIF-8-coated CeO nanoparticle Catalytic Ischemic stroke 244
2
POM nanoparticle Catalytic Ischemic stroke 245
Polypeptide-coated MnO nanoparticle Catalytic Ischemic stroke 246
2
Thioether-containing nanoparticle Non-catalytic Traumatic brain injury 251
Trimetallic nanocatalyst Catalytic Traumatic brain injury 254
Single-atom nanocatalysts of RhN , VN , and Fe-Cu-N 6 Catalytic Traumatic brain injury 258
4
4
Single-atom Mn nanocatalyst Catalytic Traumatic brain injury 259
Abbreviations: MSN: Mesoporous silica nanoparticles; POM: Polyoxometalate
al. synthesized calcium hexacyanoferrate nanoparticles Adhikari et al. prepared a citrate-modified Mn O
3
4
(KCa(H O) [Fe (CN) H O) that can mimic the activities nanoparticle and used it for treating chronic kidney
III
2
2
2
6·
of multiple antioxidases to scavenge ROS efficiently, disease. In a cisplatin-induced chronic kidney disease mice
288
inhibiting ferroptosis through the upregulation of GSH model, the nanocatalyst was demonstrated to be capable
peroxidase 4 expression, and elevating the therapeutic of restoring renal function, preserving normal kidney
efficacy of acute kidney injury. 281 architecture, ameliorating inflammation, and arresting
glomerulosclerosis. These therapeutic effects were attributed
6.2. Chronic kidney disease treatment to the maintenance of mitochondrial function and cellular
Chronic kidney disease is characterized by progressive redox homeostasis by nanocatalyst-enabled ROS scavenging.
loss of renal functions, leading to accumulation of uremic In clinics, extracorporeal blood purification (hemodialysis)
toxins that further impair the functions of other organs. is frequently used to scavenge various toxins in the blood for
282
This disease has led to a serious public health problem treating chronic kidney disease. However, during blood
289
with a prevalence of 8 – 16% worldwide. Mitochondrial purification, excessive ROS will be generated in patients,
283
ROS production is the key pathogenic mechanism of leading to severe side effect. To address this issue, Wei et al.
290
chronic kidney disease. 284-286 At present, only hemodialysis synthesized an antioxidative metal-phenolic network through
and kidney transplantation have been used in clinic the coordination between tannic acid and Cu , which can
2+
for treating the disease, but uncapable of addressing catalyze cascade antioxidative reactions to scavenge ROS.
291
oxidative stress. Antioxidant molecules, such as Vitamin The nanocatalysts were incorporated in hemoperfusion
E, N-acetylcysteine, and coenzyme Q10 have been applied microspheres and integrated in the hemodialysis device
to alleviate renal oxidative stress, but their therapeutic (Figure 19), acting as an antioxidant defense system to mitigate
efficacies are not satisfactory. 287 side effect and prevent the occurrence of complications.
Volume 1 Issue 1 (2024) 22 doi: 10.36922/imo.2527