Page 94 - IMO-2-2
P. 94
Innovative Medicines & Omics Femtomolar inhibition of pseudoeriocitrin
twice for pseudoeriocitrin (except the first docking with
SoCOX1). As a result of the repeated computations, we
obtained different values for the free binding energy at the
end of the docking process.
According to Tables 1 and 2, neither eriocitrin nor
pseudoeriocitrin could bind to any β-tubulin protein
(1OJ0, 6VAX, and EvTub) tested in this study. The
homology model and Ramachandran plot of EvTub are
shown in Figure A3. However, pseudoeriocitrin did inhibit
1OJ0 (H. contortus β-tubulin), albeit with extremely low
free binding energy. This indicates that pseudoeriocitrin
does not have a toxic effect on β-tubulin. In general,
β-tubulin inhibition is associated with the prevention
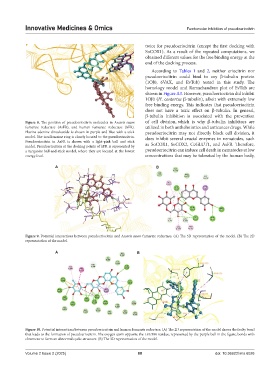
Figure 8. The position of pseudoeriocitrin molecules in Ascaris suum of cell division, which is why β-tubulin inhibitors are
fumarate reductase (AsFR), and human fumarate reductase (hFR). utilized in both anthelmintics and anticancer drugs. While
Flavine-adenine-dinucleotide is shown in purple and blue with a stick pseudoeriocitrin may not directly block cell division, it
model. The isoalloxazine ring is closely located to the pseudoeriocitrin. does inhibit several crucial enzymes in nematodes, such
Pseudoeriocitrin in AsFR is shown with a light-pink ball and stick
model. Pseudoeriocitrin at the docking points of hFR is represented by as SoCOX1, SoCOX2, CeGLUT1, and AsFR. Therefore,
a turquoise ball-and-stick model, where they are located at the lowest pseudoeriocitrin can induce cell death in nematodes at low
energy level. concentrations that may be tolerated by the human body,
A B
Figure 9. Potential interactions between pseudoeriocitrin and Ascaris suum fumarate reductase. (A) The 3D representation of the model. (B) The 2D
representation of the model.
A B
Figure 10. Potential interactions between pseudoeriocitrin and human fumarate reductase. (A) The 2D representation of the model shows the faulty bond
that leads to the formation of pseudoeriocitrin. The oxygen atom opposite the LEU306 residue, represented by the purple ball in the figure, bonds with
chromene to form an abnormal cyclic structure. (B) The 3D representation of the model.
Volume 2 Issue 2 (2025) 88 doi: 10.36922/imo.6026