Page 39 - MSAM-2-1
P. 39
Materials Science in Additive Manufacturing 3D-printed nozzle for 3D bioprinting
A B
C
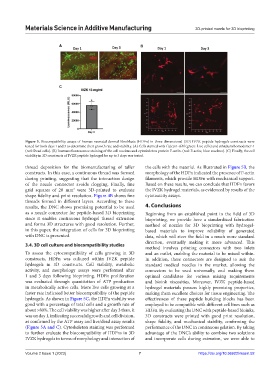
Figure 5. Biocompatibility assays of human neonatal dermal fibroblasts (HDFn) in three-dimensional (3D) IVZK peptide hydrogels constructs were
tested for both days 1 and 3 to determine their growth rate and viability. (A) Cells stained with Calcein-AM (green: Live cells) and ethidium homodimer-1
(red: Dead cells). (B) Immunofluorescence staining of the cell nucleus and cytoskeleton protein F-actin. (red: F-actin; blue: nucleus). (C) Finally, the cell
viability in 3D constructs of IVZK peptide hydrogel for up to 3 days was tested.
thread deposition for the biomanufacturing of taller the cells with the material. As illustrated in Figure 5B, the
constructs. In this case, a continuous thread was formed morphology of the HDFn indicated the presence of F-actin
during printing, suggesting that the interaction design filaments, which provide HDFn with mechanical support.
of the nozzle connector avoids clogging. Finally, fine Based on these results, we can conclude that HDFn favors
grid squares of 20 mm were 3D-printed to evaluate the IVZK hydrogel materials, as evidenced by results of the
2
shape fidelity and print resolution. Figure 4B shows fine cytotoxicity assays.
threads formed in different layers. According to these
results, the DNC shows promising potential to be used 4. Conclusions
as a nozzle connector for peptide-based 3D bioprinting Beginning from an established point in the field of 3D
since it enables continuous hydrogel thread extrusion bioprinting, we provide here a standardized fabrication
and forms 3D structures with good resolution. Further, method of nozzles for 3D bioprinting with hydrogel-
in this paper, the integration of cells for 3D bioprinting based materials to improve reliability of generated
with DNC is presented. data, which will steer the field in a much more standard
direction, eventually making it more advanced. This
3.4. 3D cell culture and biocompatibility studies
method involves printing connectors with two inlets
To assess the cytocompatibility of cells growing in 3D and an outlet, enabling the material to be mixed within.
constructs, HDFn was cultured within IVZK peptide In addition, these connectors are designed to suit the
hydrogels in 3D constructs. Cell viability, metabolic standard medical needles in the market, allowing the
activity, and morphology assays were performed after connectors to be used universally, and making them
1 and 3 days following bioprinting. HDFn proliferation optimal candidates for various mixing requirements
was evaluated through quantitation of ATP production and bioink viscosities. Moreover, IVZK peptide-based
in metabolically active cells. More live cells growing at a hydrogel materials possess highly promising properties,
faster rate indicated better biocompatibility of the peptide making them excellent choices for tissue engineering. The
hydrogels. As shown in Figure 5C, the HDFn viability was effectiveness of these peptide building blocks has been
good with a percentage of total cells and a growth rate of employed to be compatible with different cell lines such as
about >80%. The cell viability was higher after day 3 than, it HDFn. By evaluating the DNC with peptide-based bioinks,
was on day 1, indicating successful growth and cell division, 3D constructs were printed with good print resolution,
as confirmed by the ATP assay and live/dead assay results shape fidelity, and mechanical stability, confirming the
(Figure 5A and C). Cytoskeleton staining was performed performance of the DNC in continuous gelation. By taking
to further evaluate the biocompatibility of HDFns in 3D advantage of the DNC’s ability to combine two solutions
IVZK hydrogels in terms of morphology and interaction of and incorporate cells during extrusion, we were able to
Volume 2 Issue 1 (2023) 7 https://doi.org/10.36922/msam.52