Page 550 - IJB-10-4
P. 550
International Journal of Bioprinting 3D-bioprinting of osteochondral plugs
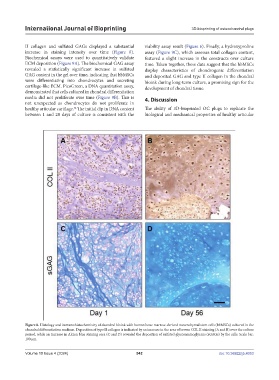
II collagen and sulfated GAGs displayed a substantial viability assay result (Figure 6). Finally, a hydroxyproline
increase in staining intensity over time (Figure 8). assay (Figure 9C), which assesses total collagen content,
Biochemical assays were used to quantitatively validate featured a slight increase in the constructs over culture
ECM deposition (Figure 9A). The biochemical GAG assay time. Taken together, these data suggest that the hbMSCs
revealed a statistically significant increase in sulfated display characteristics of chondrogenic differentiation
GAG content in the gel over time, indicating that hbMSCs and deposited GAG and type II collagen in the chondral
were differentiating into chondrocytes and secreting bioink during long-term culture, a promising sign for the
cartilage-like ECM. PicoGreen, a DNA quantitation assay, development of chondral tissue.
demonstrated that cells cultured in chondral differentiation
media did not proliferate over time (Figure 9B). This is 4. Discussion
not unexpected as chondrocytes do not proliferate in
healthy articular cartilage. The initial dip in DNA content The ability of 3D-bioprinted OC plugs to replicate the
54
between 1 and 28 days of culture is consistent with the biological and mechanical properties of healthy articular
Figure 8. Histology and immunohistochemistry of chondral bioink with human bone marrow-derived mesenchymal stem cells (hbMSCs) cultured in the
chondral differentiation medium. Deposition of type II collagen is indicated by an increase in the area of brown COL II staining (A and B) over the culture
period, while an increase in Alcian blue staining area (C and D) revealed the deposition of sulfated glycosaminoglycans (sGAGs) by the cells. Scale bar:
100 µm.
Volume 10 Issue 4 (2024) 542 doi: 10.36922/ijb.4053