Page 406 - IJB-9-6
P. 406
International Journal of Bioprinting Biofabrication for islet transplantation
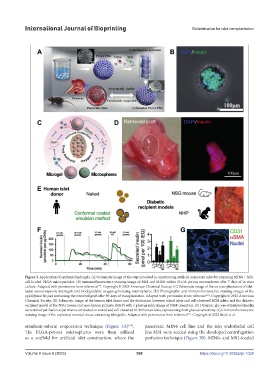
Figure 3. Application of synthesis hydrogels. (A) Schematic image of the steps involved in constructing artificial pancreatic islets by preparing MIN6 + MS1
cell-loaded PLGA microparticles. (B) Immunofluorescence staining image of MS1 and MIN6 within PLGA porous microspheres after 7 days of in vitro
culture. Adapted with permission from reference . Copyright © 2023 American Chemical Society. (C) Schematic image of the co-transplantation of islet-
[58]
laden nanocomposite microgels and biodegradable oxygen-generating microspheres. (D) Photographic and immunofluorescence staining images of the
epididymal fat pad containing the retrieved graft after 90 days of transplantation. Adapted with permission from reference . Copyright © 2022 American
[61]
Chemical Society. (E) Schematic image of the human islet donor and the distinction between naked islets and cell-clustered ECM islets, and the diabetic
recipient model of the NSG mouse and non-human primate (NHP) with a photographic image of NHP omentum. (F) Dynamic glucose-stimulated insulin
secretion of perifusion experiments conducted on naked and cell-clustered ECM human islets, representing their glucose sensitivity. (G) Immunofluorescence
staining image of the explanted omental tissue containing islet grafts. Adapted with permission from reference . Copyright © 2022 Stock et al.
[67]
[58]
emulsion-solvent evaporation technique (Figure 3A) . pancreatic MIN6 cell line and the islet endothelial cell
The PLGA-porous microspheres were then utilized line MS1 were seeded using the developed centrifugation
as a scaffold for artificial islet construction, where the perfusion technique (Figure 3B). MIN6- and MS1-loaded
Volume 9 Issue 6 (2023) 398 https://doi.org/10.36922/ijb.1024