Page 102 - AJWEP-22-6
P. 102
Li, et al.
the micropore channels of Y-zeolite. Therefore, the acid intensity and the lowest Lewis acidity for RuY-1.
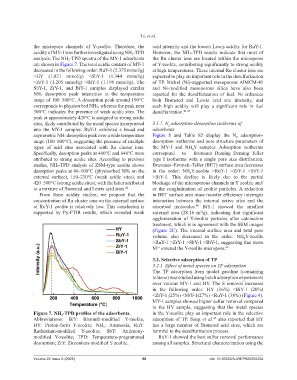
acidity of MY-1 was further investigated using NH -TPD However, the NH₃-TPD results indicate that most of
3
analysis. The NH -TPD spectra of the MY-1 adsorbents the Ru cluster ions are located within the micropores
3
are shown in Figure 7. The total acidic content of MY-1 of Y-zeolite, contributing significantly to strong acidity
decreased in the following order: RuY-1 (2.375 mmol/g) at high temperatures. These internal Ru cluster ions are
>HY (1.821 mmol/g) >SbY-1 (1.344 mmol/g) expected to play an important role in the desulfurization
>ZrY-1 (1.205 mmol/g) >BiY-1 (1.196 mmol/g). The of TP. Nickel (Ni)-supported mesoporous AlMCM-41
SbY-1, ZrY-1, and BiY-1 samples displayed similar and Ni-modified mesoporous silica have also been
NH₃ desorption peak intensities in the temperature reported for the desulfurization of fuel. Ni enhances
range of 100–500°C. A desorption peak around 190°C both Brønsted and Lewis acid site intensity, and
corresponds to physisorbed NH₃, whereas the peak near such high acidity will play a significant role in fuel
300°C indicates the presence of weak acidic sites. The desulfurization. 21,47
peak at approximately 420°C is assigned to strong acidic
sites, likely contributed by the metal species incorporated 3.1.7. N adsorption-desorption isotherms of
2
into the MY-1 samples. RuY-1 exhibited a broad and adsorbents
asymmetric NH₃ desorption peak over a wide temperature Figure 8 and Table S2 display the N adsorption-
2
range (100–800°C), suggesting the presence of multiple desorption isotherms and pore structure parameters of
types of acid sites associated with Ru cluster ions. the MY-1 and NH Y samples. Adsorption isotherms
4
Specifically, desorption peaks at 400°C and 640°C were correspond to Brunauer–Deming–Deming–Teller
attributed to strong acidic sites. According to previous type I isotherms with a single pore size distribution.
studies, NH₃-TPD analysis of ZSM-type zeolite shows Brunauer–Emmett–Teller (BET) surface area decreases
desorption peaks at 60–100°C (physisorbed NH₃ on the in the order: NH Y-zeolite >RuY-1 >ZrY-1 >SbY-1
4
external surface), 150–230°C (weak acidic sites), and >BiY-1. This decline is likely due to the partial
420–500°C (strong acidic sites), with the latter attributed blockage of the microporous channels in Y zeolite and/
to a mixture of Brønsted and Lewis acid sites. 46 or the conglutination of zeolite particles. A reduction
From these acidity studies, we propose that the in BET surface area mass transfer efficiency interrupts
concentration of Ru cluster ions on the external surface interaction between the internal active sites and the
of RuY-1 zeolite is relatively low. This conclusion is adsorbed molecules. BiY-1 showed the smallest
48
supported by Py-FTIR results, which revealed weak external area (28.16 m /g), indicating that significant
2
agglomeration of Y-zeolite particles after calcination
treatment, which is in agreement with the SEM images
(Figure 2C). The internal surface area and total pore
volume also decreased in the order: NH Y-zeolite
4
>RuY-1 >ZrY-1 >SbY-1 >BiY-1, suggesting that more
M entered the Y-zeolite micropore. 49
n+
3.2. Selective adsorption of TP
3.2.1. Effect of metal species on TP adsorption
The TP adsorption from model gasoline (containing
toluene) was studied using batch adsorption experiments
over various MY-1 and HY. The S removal increases
in the following order: HY (16%) <BiY-1 (20%)
<ZrY-1 (25%) <SbY-1(27%) <RuY-1 (38%) (Figure 9).
MY-1 samples showed higher sulfur removal compared
to the HY sample, suggesting that the metal species
Figure 7. NH -TPD profiles of the adsorbents. in the Y-zeolite play an important role in the selective
3
Abbreviations: BiY: Bismuth-modified Y-zeolite; adsorption of TP. Song et al. also reported that HY
45
HY: Proton-form Y-zeolite; NH : Ammonia; RuY: has a large number of Brønsted acid sites, which are
3
Ruthenium-modified Y-zeolite; SbY: Antimony- harmful to the desulfurization process.
modified Y-zeolite; TPD: Temperature-programmed RuY-1 showed the best sulfur removal performance
desorption; ZrY: Zirconium-modified Y-zeolite. among all samples. Structural characterization using the
Volume 22 Issue 6 (2025) 96 doi: 10.36922/AJWEP025250204