Page 103 - AJWEP-22-6
P. 103
Adsorption desulfurization
A B
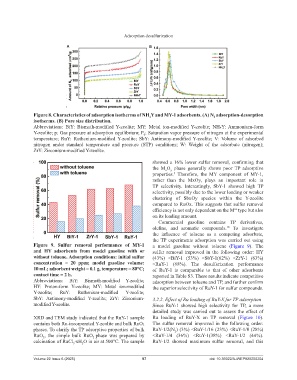
Figure 8. Characteristics of adsorption isotherms of NH Y and MY-1 adsorbents. (A) N adsorption‑desorption
2
4
isotherms. (B) Pore size distribution.
Abbreviations: BiY: Bismuth-modified Y-zeolite; MY: Metal ion-modified Y-zeolite; NH₄Y: Ammonium-form
Y-zeolite; p: Gas pressure at adsorption equilibrium; P : Saturation vapor pressure of nitrogen at the experimental
0
temperature; RuY: Ruthenium-modified Y-zeolite; SbY: Antimony-modified Y-zeolite; V: Volume of adsorbed
nitrogen under standard temperature and pressure (STP) conditions; W: Weight of the adsorbate (nitrogen);
ZrY: Zirconium-modified Y-zeolite.
showed a 16% lower sulfur removal, confirming that
the M O phase generally shows poor TP adsorptive
y
x
properties. Therefore, the MY component of MY-1,
5
rather than the MxOy, plays an important role in
TP selectivity. Interestingly, SbY-1 showed high TP
selectivity, possibly due to the lower loading or weaker
clustering of SbxOy species within the Y-zeolite
compared to RuOx. This suggests that sulfur removal
efficiency is not only dependent on the M type but also
n+
on its loading amount.
Commercial gasoline contains TP derivatives,
olefins, and aromatic compounds. To investigate
18
the influence of toluene as a competing adsorbate,
the TP experiments adsorption was carried out using
Figure 9. Sulfur removal performance of MY-1 a model gasoline without toluene (Figure 9). The
and HY adsorbents from model gasoline with or sulfur removal improved in the following order: HY
without toluene. Adsorption conditions: initial sulfur (43%) <BiY-1 (53%) <SbY-1(62%) <ZrY-1 (63%)
concentration = 20 ppm; model gasoline volume: <RuY-1 (68%). The desulfurization performance
10 mL; adsorbent weight = 0.1 g, temperature = 80°C; of RuY-1 is comparable to that of other adsorbents
contact time = 2 h. reported in Table S3. These results indicate competitive
Abbreviations: BiY: Bismuth-modified Y-zeolite; adsorption between toluene and TP, and further confirm
HY: Proton-form Y-zeolite; MY: Metal ion-modified the superior selectivity of RuY-1 for sulfur compounds.
Y-zeolite; RuY: Ruthenium-modified Y-zeolite;
SbY: Antimony-modified Y-zeolite; ZrY: Zirconium- 3.2.2. Effect of Ru loading of RuY-X for TP adsorption
modified Y-zeolite. Since RuY-1 showed high selectivity for TP, a more
detailed study was carried out to assess the effect of
XRD and TEM study indicated that the RuY-1 sample Ru loading of RuY-X on TP removal (Figure 10).
contains both Ru-incorporated Y-zeolite and bulk RuO₂ The sulfur removal improved in the following order:
phases. To clarify the TP adsorptive properties of bulk RuY-1/2(N ) (3%) <RuY-1/16 (23%) <RuY-1/8 (28%)
2
RuO , the simple bulk RuO phase was prepared by <RuY-1/4 (36%) <RuY-1(38%) <RuY-1/2 (44%).
2
2
calcination of RuCl ∙xH O in air at 500°C. The sample RuY-1/2 showed maximum sulfur removal, and this
3
2
Volume 22 Issue 6 (2025) 97 doi: 10.36922/AJWEP025250204