Page 479 - IJB-10-2
P. 479
International Journal of Bioprinting Bioprinting with ASCs and bioactive glass
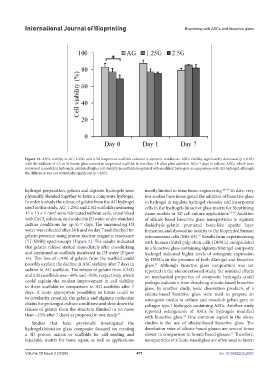
Figure 13. ASCs viability in AG, 1.25G, and 2.5G bioprinted scaffolds cultured in dynamic conditions. ASCs viability significantly decreased (p < 0.05)
with the addition of 2.5 wt.% borate glass content in bioprinted scaffolds in less than 4 h after glass addition. After 7 days in culture, ASCs, which were
recovered in modified hydrogels, exhibited higher cell viability in scaffolds bioprinted with modified hydrogels in comparison with AG hydrogel, although
the difference was not statistically significant (p < 0.05).
hydrogel preparation, gelatin and alginate hydrogels were mostly limited to bone tissue engineering. 48-54 To date, very
physically blended together to form a composite hydrogel. few studies have investigated the addition of bioactive glass
In order to study the release of gelatin from the AG hydrogel to hydrogel to regulate hydrogel viscosity and incorporate
used in this study, AG, 1.25G and 2.5G scaffolds measuring cells in the hydrogel+bioactive glass matrix for bioprinting
15 × 15 × 1 mm were fabricated without cells, crosslinked tissue models or 3D cell culture applications. 55-58 Addition
3
with CaCl solution, and soaked in DI water under standard of silicate-based bioactive glass nanoparticles to alginate
2
culture conditions for up to 7 days. The surrounding DI dialdehyde-gelatin promoted bone-like apatite layer
water was collected after 24 h and on day 7 and checked for formation and showed no toxicity to the bioprinted human
gelatin presence using proton nuclear magnetic resonance osteosarcoma cells (MG-63). Results from experimenting
55
( H-NMR) spectroscopy (Figure 2). The results indicated with human dental pulp stem cells (DPSCs) encapsulated
1
that gelatin release started immediately after crosslinking in a bioactive glass containing alginate/Matrigel composite
and continued as scaffolds incubated in DI water (Figure hydrogel indicated higher levels of osteogenic expression
14). This loss of ~70% of gelatin from the scaffold could by DPSCs in the presence of both Matrigel and bioactive
possibly explain the decline in ASC viability after 7 days in glass. Although bioactive glass composition was not
57
culture in AG scaffolds. The release of gelatin from 1.25G reported in the aforementioned study, the minimal effects
and 2.5G scaffolds was ~40% and ~50%, respectively, which on mechanical properties of composite hydrogels could
could explain the modest improvement in cell viability perhaps indicate a slow dissolving silicate-based bioactive
in these scaffolds in comparison to AG scaffolds after 7 glass. In another study, ionic dissolution products of a
days. A more appropriate possibility in future could be silicate-based bioactive glass were used to prepare an
to covalently crosslink the gelatin and alginate molecular osteogenic media to culture and crosslink gellan gum or
chains for prolonged culture conditions and slow down the collagen type I hydrogels containing ASCs. Another study
release of gelatin from the structure (limited to no more reported osteogenesis of ASCs in hydrogels modified
than ~25% after 7 days) as proposed in one study. 47 with bioactive glass. One common aspect in the above
58
Studies that have previously investigated the studies is the use of silicate-based bioactive glass. The
hydrogel+bioactive glass composite focused on creating dissolution rates of silicate-based glasses are several times
a 3D porous matrix or scaffolds for cell seeding and slower in comparison to borate-based glasses. Therefore,
15
injectable matrix for bone repair, as well as applications nanoparticles of silicate-based glass are often used to fasten
Volume 10 Issue 2 (2024) 471 doi. 10.36922/ijb.2057