Page 413 - IJB-9-6
P. 413
International Journal of Bioprinting Biofabrication for islet transplantation
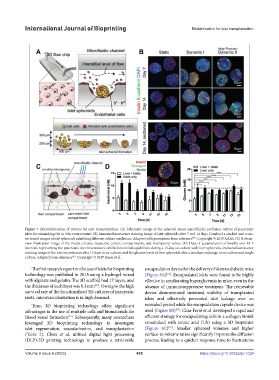
Figure 7. Microfabrication of devices for islet transplantation. (A) Schematic image of the spheroid-based microfluidic perfusion culture of pancreatic
islets for mimicking the in vivo environment. (B) Immunofluorescence staining image of islet spheroids after 7 and 14 days. Confocal z-stacked and cross-
sectioned images of islet spheroids exhibiting different culture conditions. Adapted with permission from reference . Copyright © 2019 AAAS. (C) Bottom-
[81]
view illustration image of the media circuits, respective culture compartments, and micropump valves. (D) Data of accumulation of insulin over 48-h
intervals representing the pancreatic islet microtissues exhibit functional capabilities during a 15-day co-culture with liver spheroids, immunofluorescence
staining image of the islet microtissues after 15 days in co-culture, and the glucose levels of liver spheroids after a medium exchange in co-culture and single
culture. Adapted from reference . Copyright © 2017 Bauer et al.
[83]
The first research report on the use of islets for bioprinting encapsulation device for the delivery of islets to diabetic mice
technology was published in 2015 using a hydrogel mixed (Figure 8A) . Encapsulated islets were found to be highly
[93]
with alginate and gelatin. The 3D scaffold had 17 layers, and effective in ameliorating hyperglycemia in mice, even in the
the thickness of each layer was 0.1 mm . Owing to the high absence of immunosuppressive treatment. The retrievable
[91]
survival rate of the functionalized 3D cultures of pancreatic device demonstrated sustained viability of transplanted
islets, microvascularization is in high demand. islets and effectively prevented islet leakage over an
Thus, 3D bioprinting technology offers significant extended period while the encapsulation capsule device was
[93]
advantages in the use of multiple cells and biomaterials for used (Figure 8B) . Clua-Ferré et al. developed a rapid and
blood vessel formation . Subsequently, many researchers efficient strategy for encapsulating cells in a collagen bioink
[92]
leveraged 3D bioprinting technology to investigate crosslinked with tannic acid (TA) using a 3D bioprinter
islet regeneration, vascularization, and transplantation (Figure 8C) . Smaller spheroid volumes and higher
[94]
(Table 2). Chen et al. utilized digital light processing surface-to-volume ratios significantly improve the diffusion
(DLP)-3D printing technology to produce a retrievable process, leading to a quicker response time to fluctuations
Volume 9 Issue 6 (2023) 405 https://doi.org/10.36922/ijb.1024